The bile salts are amphipathic steroid-based molecules that are synthesized in the liver using cholesterol; they are the conjugated (glycine/taurine) bile acid products.
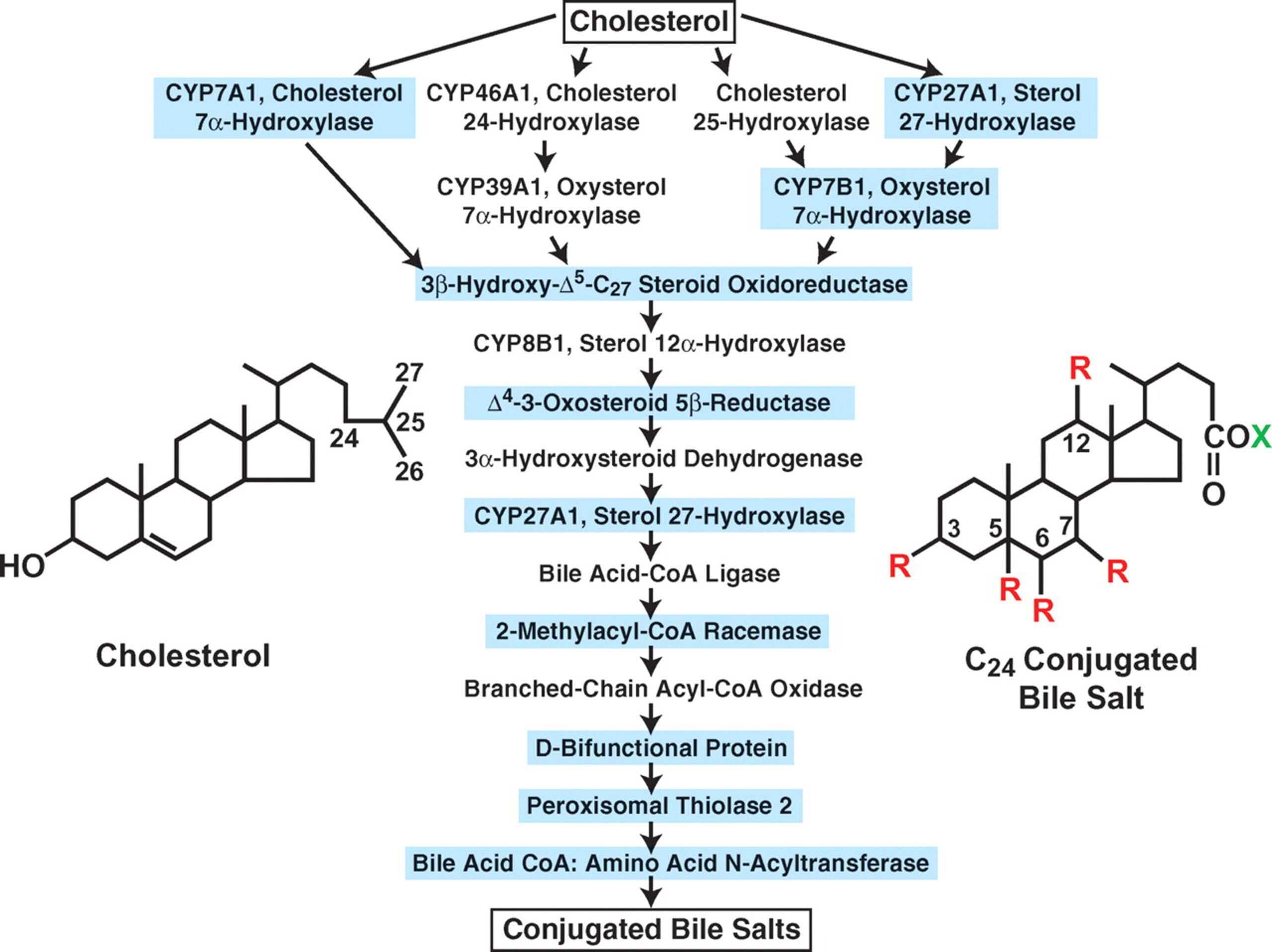
Biosynthesis starts in hepatocytes through the classical (neutral) pathway, which converts cholesterol to 7α-hydroxycholesterol catalyzed by cholesterol 7α-hydroxylase (CYP7A1), resulting in cholic acid and chenodeoxycholic acid.
- Alternative (acidic) pathway initiated by sterol 27 hydroxylase (CYP27A1) plays a major role in the formation of chenodeoxycholic acid.
- Primary bile acids are conjugated with glycine/taurine to form bile salts, which enhance solubility for biliary secretion and lipid digestion.
Site of Synthesis: The Role of Hepatocytes and Organelles
- Hepatocytes, specialized parenchymal cells that catabolize cholesterol to bile acids and then conjugate them, are the only cells in the liver that produce bile salts.
- The primary enzymes required in the biosynthesis are located within various hepatocellular organelles: the endoplasmic reticulum contains the rate-limiting cholesterol 7α hydroxylase (CYP7A1), and the mitochondria possess sterol 27 hydroxylase (CYP27A1) to oxidize the side chain.
- In peroxisomes, final side-chain shortening of side chains and conjugation of amino acids take place before exportation of bile salts into bile.
The Classic (Neutral) Pathway: The Major Route of Synthesis
- The classical pathway is the main synthesis pathway of bile acids in human beings and occurs exclusively in the liver.
- Cholesterol is initially hydroxylated at the carbon-7 site by the enzyme cholesterol 7α Hydroxylase (CYP7A1). This is the rate-limiting step in bile acid synthesis.
- The first step product, 7α-hydroxycholesterol, is further converted to 7 α -hydroxy-4-cholesten-3-one. This intermediate serves as a branching point for further processing.
- With the action of sterol 12 α hydroxylase (CYP8B1) on the intermediate, the pathway proceeds towards cholic acid (CA) production.
- CYP8B1, therefore, helps determine the hydrophilicity profile of the bile acid pool.
- The intermediate is converted to chenodeoxycholic acid (CDCA) in the absence of the activity of CYP8B1.
- Both cholic acid and chenodeoxycholic acid are synthesized in large proportions through this route and form the major portion of bile acids.
The Rate-Limiting Step: Cholesterol 7-alpha-Hydroxylase (CYP7A1)
- Cholesterol 7α-hydroxylase (CYP7A1) is the rate-limiting enzyme of the biosynthesis of bile acids in the classical pathway.
- CYP7A1 is a cytochrome P450 enzyme found primarily in the endoplasmic reticulum, which catalyzes the 7α-hydroxylation of cholesterol, committing it to the bile acid metabolism.
- Activity of CYP7A1 will define the size of the bile acid pool and the subsequent proportion of cholic acid to chenodeoxycholic acid.
- Transcriptional regulation of CYP7A1 is a major control point of hepatic bile acid production.
The Alternative (Acidic) Pathway: The Role of Mitochondrial Enzymes
- This pathway begins with the conversion of cholesterol to 27‑hydroxycholesterol by mitochondrial sterol 27-hydroxylase (CYP27A1).
- This route adds a smaller proportion (approximately 10 %) of total bile acid synthesis to the classic route.
- Since this primary hydroxylation is on the side chain but not the ring structure, the pathway can be described as an “acidic” pathway.
- The oxidized intermediate is further hydroxylated by oxysterol 7 α-hydroxylase (CYP7B1) at the 7 α position.
- Once these two primary processes have been carried out, common enzymatic processes involving the classical pathway finish the ring and side-chain remodeling to produce primary bile acids.
- Since this pathway may function in extrahepatic tissues, it offers an alternative pathway of cholesterol catabolism.
Primary Bile Acids: Formation of Cholic and Chenodeoxycholic Acid
- The first hepatic products of cholesterol catabolism are primary bile acids; the predominant ones in humans are cholic acid (CA) and chenodeoxycholic acid (CDCA).
- In the classical pathway, 7 α-hydroxycholesterol undergoes multiple reactions to produce CA and CDCA, with the ratio of CA being dictated by CYP8B1.
- The alternative route mainly produces CDCA through CYP27A1 and CYP7B1-catalyzed stages.
- These are the main bile acids, which, when conjugated, become bile salts that can be secreted into the bile.
Conjugation: Converting Bile Acids into Bile Salts (Glycine and Taurine)
- Bile acids are conjugated at the terminal carboxyl group with glycine or taurine before secretion into bile canaliculi through an amide bond.
- This reaction produces glyco- and tauro-conjugated bile acids, which enhance amphipathicity, decrease toxicity, and enhance the bile secretion.
- Conjugation takes place in 2 enzymatic phases:
- Bile acid-CoA synthetase (BACS), encoded by SLC27A5, activates bile acids by forming bile acid-CoA thioesters.
- This is then followed by the attachment of glycine or taurine by bile acid-CoA: amino acid N-acyltransferase (BAAT).
- Both primary (CA, CDCA) and secondary (DCA, LCA) bile acids serve as substrates.
- SLC27A5 and BAAT genes are mainly liver-expressed genes and are controlled by the nuclear receptor, FXR.
- Glycocholic acid (GCA), taurocholic acid (TCA), and other derivatives conjugated to glycine and taurine are common human bile salts.
Regulation of Biosynthesis: Negative Feedback Loops and FXR
- Bile salt (bile acid) synthesis is tightly regulated by a negative feedback mechanism to prevent the accumulation of toxic bile acid metabolites.
- When intracellular bile acid levels rise, FXR activation initiates negative feedback to prevent overproduction. It suppresses transcription of the rate-limiting enzyme CYP7A1, thereby reducing bile acid synthesis.
- The Farnesio X Receptor (FXR) is a nuclear receptor that is highly expressed in the liver and intestine and is a receptor that detects the levels of bile acids and maintains bile acid homeostasis.
- Chenodeoxycholic acid (CDCA) is the strongest natural FXR agonist, and cholic acid (CA) is a weaker agonist.
- FXR triggers small heterodimer partner (SHP) and fibroblast growth factor 19 (FGF19) signaling on activation by bile acids that inhibit CYP7A1 and CYP8B1 expression, decreasing bile acid production.
Defects in Biosynthesis: Cerebrotendinous Xanthomatosis and Genetic Errors
Cerebrotendinous xanthomatosis
- Cerebrotendinous xanthomatosis (CTX) is a rare autosomal recessive disorder of bile acid production because of impaired cholesterol metabolism.
- It is caused by a mutation in the CYP27A1 gene that encodes the mitochondrial sterol 27-hydroxylase involved in the production of bile acids.
- Mutation leads to loss of production of chenodeoxycholic acid and the accumulation of cholestanol and cholesterol in the tissues, especially those of the brain, tendons, and the lens.
- It is manifested clinically by tendon xanthomas, juvenile cataracts, chronic diarrhea, neurological dysfunction, and premature atherosclerosis.
- Biochemical abnormalities can be improved through early diagnosis and replacement with chenodeoxycholic acid.
Genetic errors
- Genetic defects in bile salt production affect the last stages in the synthesis of conjugated bile salts out of primary bile acids.
- Mutations in SLC27A5 (Bile acid-CoA ligase) inhibit the conversion of the free bile acids to bile acid-CoA, which is required in the subsequent conjugation.
- The mutations in BAAT (Bile acid-CoA: amino acid N-acyltransferase) block the conjugation of activated bile acids with glycine or taurine, the last step of conjugation.
- These defects cause a drop in the pool of functional bile salts, which causes the accumulation of unconjugated bile acids and hinders the formation of the micelles, hence diminishing the absorption of fats and fat-soluble vitamins.
Summary of Key Enzymes and Co-factors involved
- Cholesterol 7alpha-hydroxylase (CYP7A1): The CYP7A1 cytochrome P450 enzyme is the rate-limiting enzyme that hydroxylates cholesterol at C 7 to start the classical bile acid pathway in hepatocytes.
- 3β‑hydroxy‑Δ⁵‑C₂₇‑steroid oxidoreductase (HSD3B7): Converts the 3β‑hydroxyl group to a 3‑oxo intermediate and epimerizes it, an important initial step in bile acid synthesis.
- Sterol 12α‑hydroxylase (CYP8B1): Regulates the production of cholic acid over chenodeoxycholic acid through the incorporation of a 12α‑hydroxyl group.
- Secondary hydroxylases (e.g., sterol 27-hydroxylase, CYP27A1) participate in both classic and alternative (acidic) pathways.
- Bile acid-CoA synthetase (BACS/SLC 27A5) catalyzed the conversion of bile acids to CoA thioesters.
- Bile acid-CoA: amino acid N-acyltransferase (BAAT) pairs the activated acids with glycine or taurine to produce water-soluble bile salts.
- NADPH and O2 are necessary to carry out hydroxylation reactions by the P450 enzymes.
- CoA, glycine, taurine: These are essential in the activation and conjugation of bile acids so that they can be soluble and functional in the emulsification of fats.
Conclusion
- Bile salts are steroid-derived amphipathic molecules that are produced exclusively in hepatocytes using cholesterol and are necessary in the digestion of lipids and the catabolism of cholesterol.
- The principal pathway of biosynthesis is the classical (neutral) route in the liver, catalyzed by cholesterol 7alpha-hydroxylase (CYP7A1), the rate-limiting enzyme involved in the biosynthesis of bile acid from cholesterol.
- The alternative route, activated by mitochondrial CYP27A1, is a lesser proportion of bile acid contributors and is mainly involved in the production of chenodeoxycholic acid (CDCA).
- The two main primary bile acids produced are cholic acid (CA) and CDCA; CYP8B1 regulates the ratio of CA to CDCA and affects the bile acid hydrophilicity.
- Through BACS (SLC27A5) and BAAT, bile acids are conjugated with either glycine or taurine, which increases solubility, lowers toxicity, and allows biliary secretion and micelle formation.
- The biosynthesis of this process is tightly controlled by negative feedback via the nuclear receptor FXR, in which bile acids (particularly CDCA) inhibit CYP7A1 and CYP8B1 to ensure bile acid homeostasis.
- Malfunction of the production of bile salts is caused by genetic defects, like CYP27A1 mutations that cause cerebrotendinous xanthomatosis or an error in conjugation enzymes and result in lipid malabsorption and systemic complications.
References
- Chiang, J. Y. L., & Ferrell, J. M. (2022). Discovery of farnesoid X receptor and its role in bile acid metabolism. Molecular and cellular endocrinology, 548, 111618. https://doi.org/10.1016/j.mce.2022.111618
- King, M. W. (n.d.). Bile acid synthesis, metabolism, and biological functions. The Medical Biochemistry Page.
- M. F. (2015). A rare neurodegenerative disorder with characteristic neuroimaging findings: Cerebrotendinous xanthomatosis. Translational Research, 176(1), 1-8
- Pellicoro, A., & Faber, K. N. (2007). Review article: The function and regulation of proteins involved in bile salt biosynthesis and transport. Alimentary pharmacology & therapeutics, 26 Suppl 2, 149–160. https://doi.org/10.1111/j.1365-2036.2007.03522.x
- Sundaram, S. S., Bove, K. E., Lovell, M. A., & Sokol, R. J. (2008). Mechanisms of disease: Inborn errors of bile acid synthesis. Nature Clinical Practice. Gastroenterology & hepatology, 5(8), 456–468. https://doi.org/10.1038/ncpgasthep1179
- Tuscany Diet source (2016). Bile salts. https://www.tuscany-diet.net/2016/08/14/bile-salts/
