Bile acids are specific and quantitatively important organic components of bile, which are synthesized by hepatocytes from cholesterol.
The bile acids are amphipathic in nature since they possess both polar and nonpolar groups.
- The bile acids possess 24 carbon atoms, 2 or 3 hydroxyl groups in the steroid nucleus, and a side chain ending in a carboxyl group.
- They serve as emulsifying agents in the intestine and actively participate in the digestion and absorption of lipids.
Structure and Chemical Properties of Bile Acids
Structure of Bile Acids
- Bile acids are steroid molecules formed by cholesterol and have a hard nucleus of four fused rings (three six-membered and one five-membered).
- Hydroxyl (-OH) groups are attached to the specific positions in a or b orientation, providing a specific 3D shape.
- The hydrocarbon side chain ends with a carboxyl (-COOH) group, and this makes them amphipathic.
Chemical Properties of Bile Acids
- The bile acids are amphipathic with a hydrophilic face (hydroxyl and carboxyl groups) and a hydrophobic face (steroid rings).
- The carboxyl group is acidic, and its pKa is approximately 5-6, which enables it to be ionized at intestinal pH to be soluble.
- Conjugation to glycine or taurine reduces pKa, increasing aqueous solubility and micelle formation.
- The less hydrophilic nature of the secondary bile acids is due to the reduced number of hydroxyl groups, which influences their detergent effect and signaling capacity.
- The amphipathic nature underlies their detergent activity, emulsification of lipids, and interaction with specific receptors to regulate metabolic pathways.
Biosynthesis of Bile Acids: The Classical and Alternative Pathways
- The synthesis of bile acids is the major pathway of cholesterol catabolism in mammals.
- Bile acids are synthesized from cholesterol in the liver through two pathways: the classic pathway and the alternative pathway.
The classical (neutral) pathway
- The classical pathway is the main synthesis pathway of bile acids in human beings and occurs exclusively in the liver.
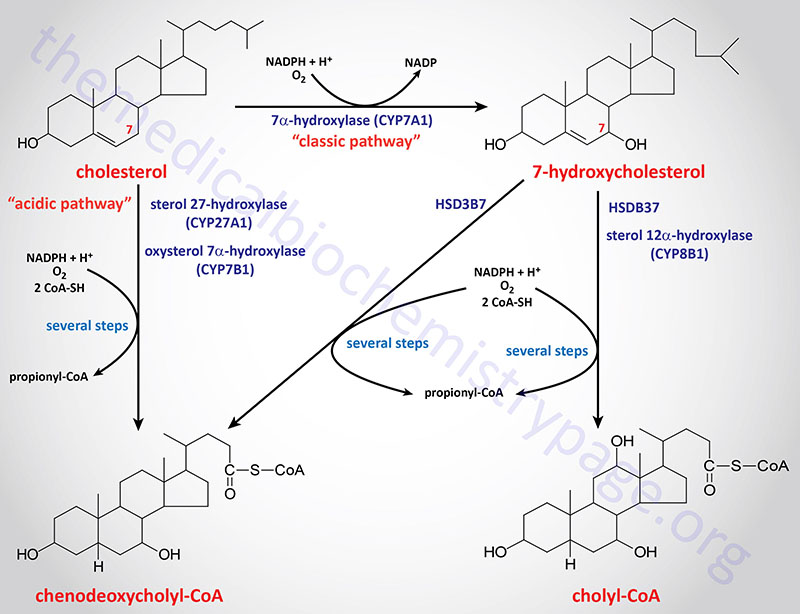
- Cholesterol is initially hydroxylated at the carbon-7 site by the enzyme cholesterol 7α-Hydroxylase (CYP7A1). This is the rate-limiting step in bile acid synthesis.
- The first step product, 7α-hydroxycholesterol, is further converted to 7 α -hydroxy-4-cholesten-3-one. This intermediate serves as a branching point for further processing.
- With the action of sterol 12 α hydroxylase (CYP8B1) on the intermediate, the pathway proceeds towards cholic acid (CA) production.
- CYP8B1, therefore, helps determine the hydrophilicity profile of the bile acid pool.
- The intermediate is converted to chenodeoxycholic acid (CDCA) in the absence of the activity of CYP8B1.
- Both cholic acid and chenodeoxycholic acid are synthesized in large proportions through this route and form the major portion of bile acids.
Alternative (Acidic) Bile Acid Synthesis Pathway
- A secondary pathway is a non-major biosynthetic pathway in humans, which only contributes a minor proportion of the total bile acid production compared to the classical pathway.
- Cholesterol is hydroxylated at the C-27 position in the first step, which is catalyzed by sterol 27-hydroxylase (CYP27A1).
- Since this primary hydroxylation is on the side chain but not the ring structure, the pathway can be described as an “acidic” pathway.
- The oxidized intermediate is further hydroxylated by oxysterol 7 α-hydroxylase (CYP7B1) at the 7 α position.
- Once these two primary processes have been carried out, common enzymatic processes involving the classical pathway finish the ring and side-chain remodeling to produce primary bile acids.
- Although quantitatively minor in adult humans, the acidic pathway is physiologically relevant (e.g., in neonates or under certain metabolic conditions).
Key distinction: The classical pathway starts at C-7 on cholesterol, but the alternative one starts at C-27 and oxidizes the side chain.
Primary vs. Secondary Bile Acids: Understanding the Difference
Primary bile acids
- Primary bile acids are those bile acids that are produced by hepatocytes (liver cells).
- The two most important primary bile acids in human beings are:
- Cholic acid
- Chenodeoxycholic acid
- Cholesterol is the main precursor to primary bile acid synthesis, which occurs via a series of oxidation events with the involvement of cytochrome P450 enzymes.
- The rate-limiting step in primary bile acid synthesis is the addition of a hydroxyl group at the 7α-position of the steroid nucleus by the enzyme cholesterol 7α-hydroxylase.
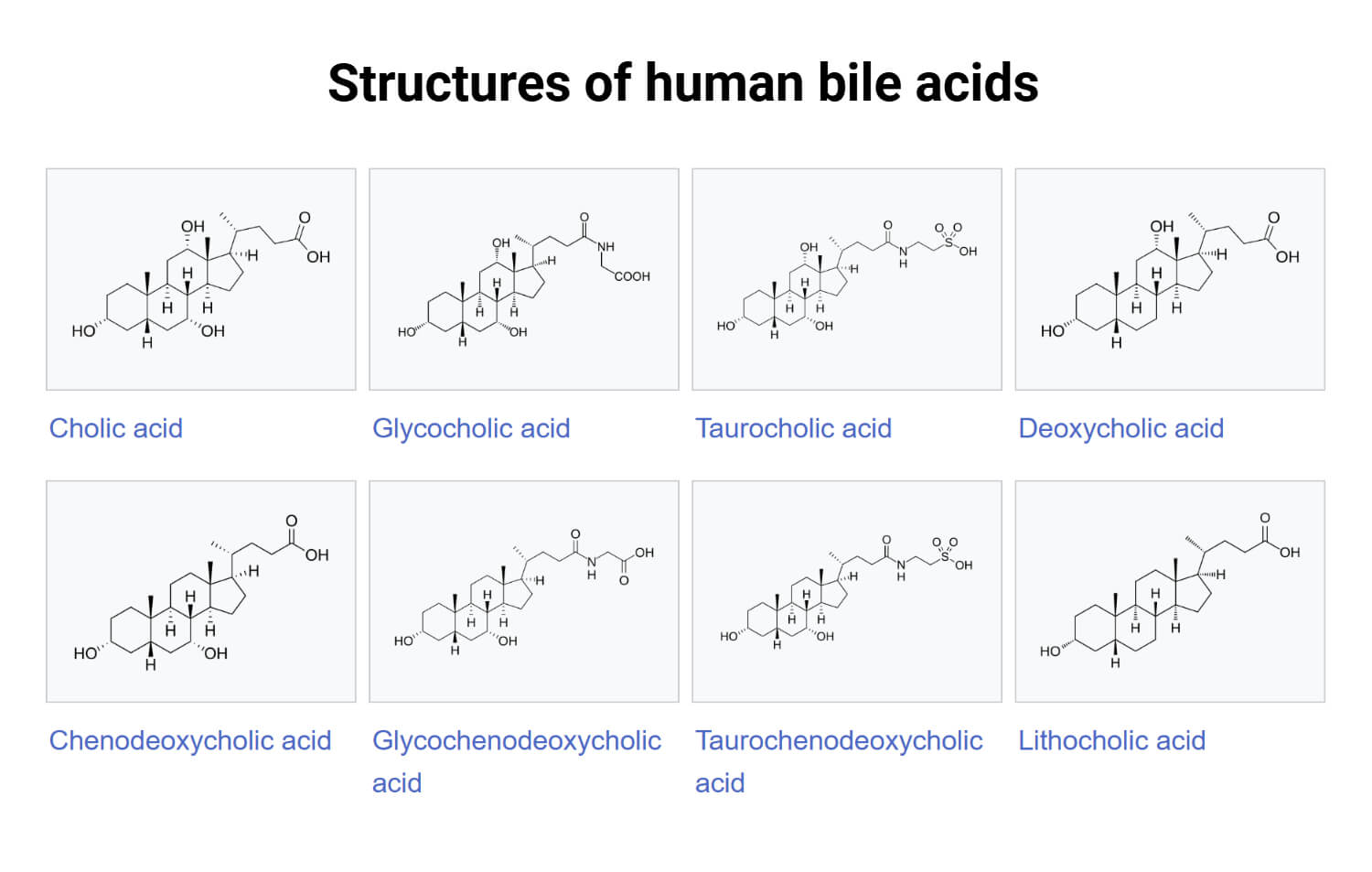
- Once it is synthesized, primary bile acids are usually conjugated with either taurine or glycine in the liver to produce bile salts (e.g., taurocholic acid, glycocholic acid, taurochenodeoxycholic acid, glycochenodeoxycholic acid).
- The conjugation enhances the bile acid water solubility and reduces cytotoxicity, promoting their ability to digest fats.
Secondary bile acids
- Secondary bile acids are not produced in the liver but rather are produced in the intestinal tract due to the activity of gut bacteria on primary bile acids.
- Secondary bile acids are not produced in the liver but rather are produced in the intestinal tract due to the activity of gut bacteria on primary bile acids.
- As the primary bile acids reach the small intestine, colonic bacteria partially dehydroxylate (lose a hydroxyl group at the 7-α position).
- This bacterial transformation converts:
- Cholic acid → Deoxycholic acid
- Chenodeoxycholic acid → Lithocholic acid
- These metabolites (deoxycholic acid and lithocholic acid) of bacteria are the main secondary bile acids present in human bile.
- Like primary bile acids, secondary bile acids may also be conjugated in glycine or taurine to give other bile salts in bile.
The Role of Gut Microbiota: Deconjugation and 7-Alpha-Dehydroxylation
- Gut microbiota controls the metabolism of bile acids by converting the primary bile acids that enter the colon.
- Intestinal bacteria synthesize bile salt hydrolases (BSHs) that deconjugate taurine-conjugated cholic acid (T-CA), glycine-conjugated cholic acid (G-CA), taurine-conjugated chenodeoxycholic acid (T-CDCA), and glycine-conjugated chenodeoxycholic acid (G-CDCA) into free bile acids.
- After the process of deconjugation, cholic acid (CA) and chenodeoxycholic acid (CDCA lose the 7-hydroxyl group, and this is performed by bacterial 7α-dehydroxylase.
- This reaction produces the secondary bile acids deoxycholic acid (DCA) from CA and lithocholic acid (LCA) from CDCA.
- The majority of LCA is excreted, and a minor amount of DCA is reabsorbed and enters the bile acid pool.
- These microbial enzymes control bile acid composition, pool size, and metabolic homeostasis.
Conjugation of Bile Acids: Glycine and Taurine Ratios
- Bile acids, which are newly synthesized, are conjugated in the liver at the terminal carboxyl group with glycine or taurine to produce glyco- and tauro-bile acids, respectively.
- This conjugation enhances hydrophilicity, decreases pKa of the side chain, decreases toxicity, and increases secretion into bile.
- The bile acids are first activated to bile acid-CoA in hepatocytes by bile acid-CoA synthetase and subsequently conjugated to glycine or taurine by bile acid-CoA: amino acid N-acyltransferase.
- The normal human ratio of glycine to taurine conjugates of bile acids is 3:1.
- The ratio of the glycine/taurine conjugates may be altered under the influence of alimentary and hormonal factors in some liver diseases.
Enterohepatic Circulation: The Recycling Mechanism of the Body
- The continuous recycling of bile acids between the liver, intestine, and portal circulation is known as Enterohepatic circulation (EHC), which keeps the homeostasis of the bile acids.
- The bile acids released in the intestine facilitate the digestion of fats and fat-soluble vitamins.
- In the terminal ileum, conjugated bile acids are reabsorbed through the Apical Sodium-Dependent Bile Acid Transporter (ASBT), which reabsorbs approximately 95% of conjugated bile acids.
- The reabsorbed bile acids leave enterocytes by Organic Solute Transporter alpha and beta (OSTa/OSTb) into portal blood.
- They are absorbed by hepatocytes via the Na + -dependent Taurocholate Co-transporting Polypeptide (NTCP) and resecrete them into bile primarily through the Bile Salt Export Pump (BSEP).
- The de novo synthesis of bile acids in the liver replaces approximately 5 % of the amount lost in feces.
Physiological Functions: Emulsification and Lipid Absorption
- The conjugated bile acids are amphipathic surfactants that have hydrophobic and hydrophilic surfaces that interact at the lipid-water interface.
- The amphipathic properties of these allow the emulsification of dietary fats, which breaks up large lipid droplets into small droplets.
- Emulsification does not involve the breaking of the lipids chemically, but helps anchor the pancreatic lipase and colipase cofactor at the water-oil interface to allow effective hydrolysis.
- The bile acids arrange lipolytic products (fatty acids, monoglycerides, cholesterol) into micelles and increase the solubility in the aqueous lumen of the intestines.
- Micelles carry lipids to the enterocyte brush border, allowing uptake and absorption.
- In the absence of bile acids, the ability to absorb lipids would be lower, as dietary fats that are insoluble in water are less available to digestive enzymes.
Bile Acids as Signaling Molecules: FXR and TGR5 Receptors
- In addition to being used as detergents, bile acids are also endogenous signaling molecules that control metabolism by binding to receptors.
- The Farnesio X Receptor (FXR) is a nuclear receptor that is highly expressed in the liver and intestine and is a receptor that detects the levels of bile acids and maintains bile acid homeostasis.
- Chenodeoxycholic acid (CDCA) is the strongest natural FXR agonist, and cholic acid (CA) is a weaker agonist.
- FXR triggers small heterodimer partner (SHP) and fibroblast growth factor 19 (FGF19) signaling on activation by bile acids that inhibit CYP7A1 and CYP8B1 expression, decreasing bile acid production.
- Takeda G-protein-coupled receptor 5 (TGR5: GPBAR1) is a membrane receptor that is stimulated by secondary bile acids, including deoxycholic acid (DCA) and lithocholic acid (LCA).
- TGR5 stimulation causes cAMP signals, which maximize energy metabolism, increase GLP-1 release by enteroendocrine cells, stimulate insulin release, and decrease inflammation.
Clinical Significance: Cholestasis and Bile Acid Malabsorption
Cholestasis
- Cholestasis is a condition that is defined by decreased or impaired bile flow of the liver to the small intestine.
- It leads to the hepatic build-up of bile acids, bilirubin, and cholesterol, resulting in hepatocellular and bile duct damage.
- Etiology involves intrahepatic (hepatocellular dysfunction, genetic transporter defects) and extrahepatic obstruction of the bile ducts.
- Clinical implications are malabsorption of fat and fat-soluble vitamins, pruritus, jaundice, and hepatic destruction.
Bile Acid Malabsorption
- Bile acid malabsorption is a condition that arises as a result of insufficient reabsorption of bile acids in the terminal ileum.
- The extra bile acids will pass into the colon, and they prompt the release of water and electrolytes.
- It results in chronic watery diarrhea, urgency, and abdominal discomfort.
- The causes involve ileal disease, ileal resection, or a dysbalance in bile acid production.
Therapeutic Applications: Ursodeoxycholic Acid (UDCA) and Chenodiol
Ursodeoxycholic Acid (UDCA)
- UDCA is a hydrophilic bile acid that is used to treat cholestatic liver disease, such as primary biliary cirrhosis and other liver cholestatic diseases.
- It protects cholangiocytes and hepatocytes against cytotoxic hydrophobic bile acids and enhances bile flow (choleresis).
- UDCA will alter the bile acid pool, reducing toxic bile acids, decreasing apoptosis, and facilitating bile acid excretion.
- It also lowers cholesterol saturation in bile and may facilitate the dissolution of the gallstones.
Chenodiol
- Chenodiol is a naturally occurring bile acid that is used medically to dissolve cholesterol gallstones in patients with a functional gallbladder who are unable to undergo surgery.
- Chenodiol decreases hepatic cholesterol production and decreases bile cholesterol saturation following oral absorption and hepatic uptake.
- It lowers cholesterol saturation, thereby facilitating the dissolution of the existing cholesterol gallstones, especially small floating stones.
- Long-term treatment (≥2 years) has the potential to dissolve stones in some patients.
Conclusion
- Bile acids are cholesterol-derived amphipathic molecules needed in lipid digestion, absorption, and catabolism of cholesterol.
- Their structure and conjugation with glycine or taurine increase solubility, decrease toxicity, and increase the formation of micelles.
- Primary bile acids are produced in the liver by classical and alternative pathways, and secondary bile acids are produced by gut microbial modification.
- Enterohepatic circulation maintains the homeostasis of bile acids; approximately 95 percent of bile acids are reabsorbed in the ileum.
- Beyond digestion, bile acids act as signaling molecules (FXR, TGR5) regulating metabolism, and their dysregulation underlies cholestasis and bile acid malabsorption.
References
- Berg, J. M., Tymoczko, J. L., Gatto, G. J., Jr., & Stryer, L. (2002). Biochemistry (5th ed.). In Molecular biology of the cell [NCBI Bookshelf].
- Cai, S. Y., & Boyer, J. L. (2021). The role of bile acids in cholestatic liver injury. Annals of translational medicine, 9(8), 737. https://doi.org/10.21037/atm-20-5110
- Chiang, J. Y. L., & Ferrell, J. M. (2020). Bile acid receptors FXR and TGR5 signaling in fatty liver diseases and therapy. American journal of physiology. Gastrointestinal and liver physiology, 318(3), G554–G573. https://doi.org/10.1152/ajpgi.00223.2019
- di Gregorio, M. C., Cautela, J., & Galantini, L. (2021). Physiology and Physical Chemistry of Bile Acids. International journal of molecular sciences, 22(4), 1780. https://doi.org/10.3390/ijms22041780
- Ramakrishnan, N., & Shah, P. (2023). Physiology, bile acids. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK549765/
- Shulpekova, Y., Zharkova, M., Tkachenko, P., Tikhonov, I., Stepanov, A., Synitsyna, A., Izotov, A., Butkova, T., Shulpekova, N., Lapina, N., Nechaev, V., Kardasheva, S., Okhlobystin, A., & Ivashkin, V. (2022). The Role of Bile Acids in the Human Body and in the Development of Diseases. Molecules (Basel, Switzerland), 27(11), 3401. https://doi.org/10.3390/molecules27113401
- Stan, S. I., Biciuşcă, V., Clenciu, D., Mitrea, A., Boldeanu, M. V., Durand, P., & Dănoiu, S. (2024). The therapeutic mechanisms and beneficial effects of ursodeoxycholic acid in the treatment of nonalcoholic fatty liver disease: a systematic review. Medicine and pharmacy reports, 97(1), 12–25. https://doi.org/10.15386/mpr-2629
- Tymoczko, J. L., Berg, J. M., & Stryer, L. (2025, September 5). Bile acid synthesis, metabolism, and biological functions. The Medical Biochemistry Page. https://themedicalbiochemistrypage.org/bile-acid-synthesis-metabolism-and-biological-functions/
- WebMD. (n.d.). Chenodiol (Chenodal, Ctexli, Chenix): Uses, side effects, interactions, pictures, warnings & dosing.
