Trypan blue staining is a fundamental technique in cell biology that uses dye exclusion to quickly assess and differentiate viable (live) from non-viable (dead or compromised) cells within a population, typically in a cell culture suspension.
It’s often used during routine cell culture maintenance, cell counting with a hemacytometer, and in cytotoxicity studies.
The very first use of trypan blue stain was carried out by, or rather, the technique was a pioneering work of the scientist Paul Ehrlich. The stain was synthesized in 1904. Its original purpose was as a potential chemotherapeutic agent to treat parasitic diseases.
While Trypan blue did not achieve widespread success as a therapeutic drug, its primary role today in cell biology emerged from the realization of its selective affinity for compromised cells.
By the early 20th century, scientists had formalized this observation into the dye exclusion test, which relies on the ability of the dye to penetrate only cells with damaged membranes. This discovery laid the foundation for its modern, routine application in cell viability assays. The first noted formal description of using Trypan blue specifically for analysing cell viability is generally credited to A. M. Pappenheimer in the mid-20th century.
Principle of Trypan Blue Staining
The effectiveness of Trypan blue staining rests entirely on the cell’s membrane integrity, a mechanism known as the dye exclusion principle.
Trypan blue is classified as an acid azo dye, specifically a diazo dye (containing two azo groups, -N=N-) derived from toluidine. It is a relatively large molecule, with a molecular weight of approximately 960.8g/mol.
The critical features that explain Trypan blue’s use in the dye exclusion test are its large size and its strong polarity/charge
The molecule contains four sulfonate SO groups (as the tetrasodium salt). These groups make the molecule highly hydrophilic (water-soluble) and confer a strong negative charge at physiological pH
This combination of large size and high negative charge prevents the dye from passively diffusing across a healthy cell’s plasma membrane, which is a lipid bilayer (hydrophobic) with an electrical potential. The intact membrane’s selective barrier actively excludes the dye.
When a cell dies or its viability is compromised, its cell membrane loses its structural and functional integrity, becoming porous or leaky. This breach allows the large, charged Trypan blue molecule to pass through the membrane and enter the cytoplasm. Once inside, the dye binds non-covalently to intracellular proteins, resulting in the distinctive blue colour observed in non-viable cells.
There are several dyes that can easily enter dead cells but are excluded by live cells. Trypan blue at a concentration of 200-400 μg/ml is taken up by dead cells at pH 7.2 to 7.5.
Other dyes like TTC (2, 3, 5 triphenyl tetrazolium chloride) and MTT (Methyl thiazole tetrazolium) can also be used for viability.
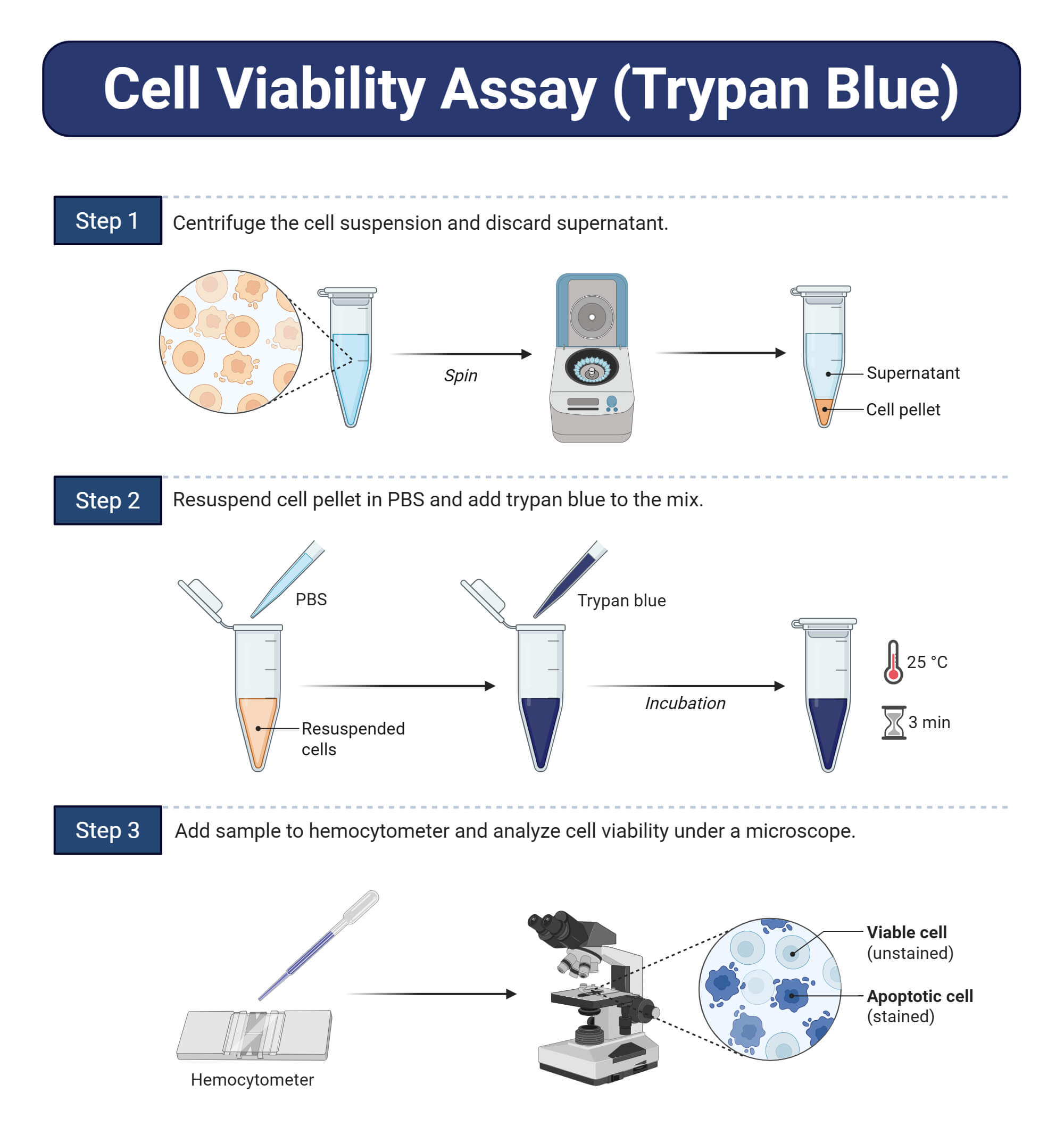
Protocol of Trypan Blue Staining
- Clean the haemocytometer chamber, dry it with alcohol, and wipe it with a clean muslin cloth. Mount the cover slips in the mounting chamber.
- To 0.9ml of cell suspension, add 0.1ml of 4% trypan blue, mix it, and allow it to stand for 5 minutes.
- With the cover slip in place, transfer a small amount of trypan blue cell suspension to both chambers of the haemocytometer, carefully touching the edge of the cover slip with the pipette tip and allowing each chamber to fill by capillary action. Do not overflow the chamber.
- Focus the haemocytometer under the microscope first at low power (10X) and then at high power (40X).
- Count all the cells in the 4 WBC chamber. Omit those cells lying on the lines of the top and left-hand side, but include those on the lines below and right-hand side.
- Determine the cell count/ml. Adjust the count to 1 x 106 / ml.
- Count the number of live (unstained) and dead (stained) cells and calculate the percent viability of cells.
The general formula for Cell viability is:
Cell Viability % = Number of Viable Cells\Total Number of Cells X 100
Where:
- Number of Viable Cells is the count of unstained (live) cells.
- Total Number of Cells is the count of both unstained (live) and stained (dead) cells.
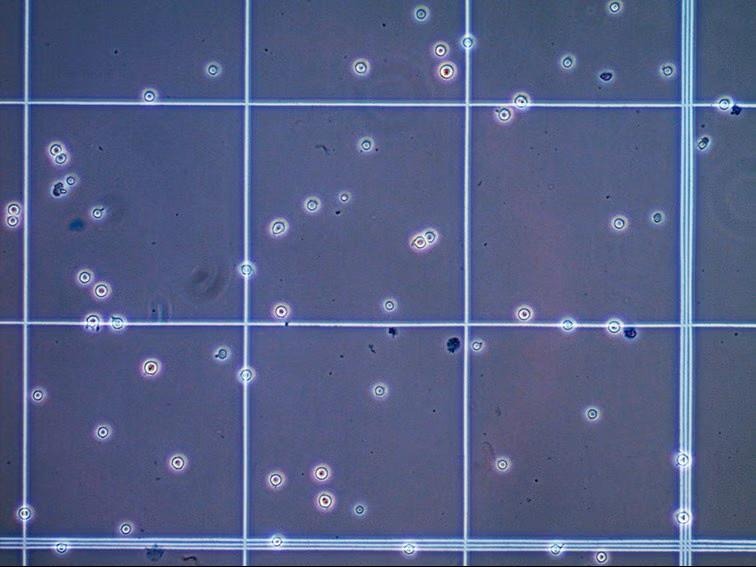
The image shows cells viewed on a hemacytometer grid after Trypan Blue staining. Viable cells appear bright or refractile (clear/unstained) with a distinct, often circular, shape. Dead cells appear blue or darkly stained because the dye has entered their cytoplasm through the compromised membrane.
https://www.sigmaaldrich.com/NP/en/technical-documents/technical-article/cell-culture-and-cell-culture-analysis/imaging-analysis-and-live-cell-imaging/cell-viability-and-proliferation
Applications of Trypan Blue Staining
The most frequent application of Trypan Blue is for the routine maintenance and health monitoring of cell cultures.
Before starting an experiment, a known number of viable cells must be plated. Trypan Blue allows researchers to accurately count the concentration of live cells (cells/mL) in the stock culture and calculate the volume needed to achieve a specific seeding density.
Cells often suffer damage during the freezing and thawing process. Trypan Blue is immediately used after thawing to determine the viability, helping researchers decide if the recovered cell population is healthy enough for use or needs further recovery.
By testing a range of concentrations of a test substance, Trypan Blue helps establish the relationship between the dose and the degree of cell death.
Trypan Blue staining is used to quantify the percentage of cells that are non-viable (killed) following exposure to a drug, compound, or environmental toxin. The resulting viability data is crucial for calculating parameters such as the IC 50 (half-maximal inhibitory concentration), which measures the effectiveness of a substance in inhibiting a biological or biochemical function.
For assays like Western blotting, ELISA, or flow cytometry, researchers often require the input to be normalized to the concentration of viable cells, not just the total cell mass. Trypan Blue provides the reliable live cell count needed for this normalization.
In isolating primary cells (e.g., from tissue samples or blood), the suspension often contains significant debris and dead cells. Trypan Blue is used to check the viability of the isolated fraction before continuing with culturing or further analysis
Trypan Blue is utilized to stain fungal hyphae and other microbial structures within plant tissues, thereby improving their visibility under a microscope to facilitate the study of host-pathogen interactions.
Advantages of Trypan Blue Staining
The assay is exceptionally fast and simple to perform. It requires minimal preparation with basic laboratory equipment: a light microscope and a hemacytometer. This makes it an ideal method for routine, high-volume checks during daily cell culture work, where time efficiency is critical
It is highly cost-effective compared to modern alternatives. Fluorescent dyes (like Propidium Iodide or Calcein AM) and automated cell counters require expensive reagents and specialized, high-capital equipment
The assay provides a direct, quantitative count of both viable (unstained) and non-viable (stained) cells. This yields an absolute viability percentage
Disadvantages of Trypan Blue Staining
It fails to assess the cell’s metabolic health (its ability to perform basic functions or proliferate). A cell that is dying slowly or is metabolically compromised may still exclude the dye and be incorrectly scored as “live.
The assay generally detects necrosis (accidental cell death involving membrane rupture) but is poor at detecting the early stages of apoptosis (programmed cell death), which often maintains membrane integrity until the very final stages.
If cells are clumped together, distinguishing individual live and dead cells becomes nearly impossible, leading to imprecise counts
References
- Cell Viability and Proliferation Assays
- Strober, W. (2015). Trypan blue exclusion test of cell viability. Current Protocols in Immunology, 111(A3.B), A3.B.1–A3.B.3. https://doi.org/10.1002/0471142735.ima03bs111
- Creative Bioarray. (n.d.). Trypan blue staining assay. Retrieved from https://www.creative-bioarray.com/support/trypan-blue-staining-assay.htm
- https://www.bing.com/ck/a?!&&p=78dbec0bb7c5647f3c26a24802dc1869d5c8695892e8c7a828cbd9cc68afbfb3JmltdHM9MTc2MTQzNjgwMA&ptn=3&ver=2&hsh=4&fclid=2b678930-794b-6467-30df-9f45784a655c&u=a1L3ZpZGVvcy9yaXZlcnZpZXcvcmVsYXRlZHZpZGVvP3E9dHJ5cGFuK2JsdWUrYXNzYXkmbWlkPTUyODRDQjY5NkJDRUNDRkUyRDc4NTI4NENCNjk2QkNFQ0NGRTJENzgmRk9STT1WSVJF
- https://www.bing.com/ck/a?!&&p=c67de7d6ab08fd41ee5c70de7f82450d5af2ca35f8d79008cc85456c25ae782dJmltdHM9MTc2MTQzNjgwMA&ptn=3&ver=2&hsh=4&fclid=2b678930-794b-6467-30df-9f45784a655c&u=a1L3ZpZGVvcy9yaXZlcnZpZXcvcmVsYXRlZHZpZGVvP3E9dHJ5cGFuK2JsdWUrYXNzYXkrcHJvdG9jb2wmbWlkPTI0QTkwMzdFRkQ3NTJCMDUwM0FFMjRBOTAzN0VGRDc1MkIwNTAzQUUmRk9STT1WSVJF
