The metabolic pathway through which fatty acids are esterified to a backbone of glycerol to produce triacylglycerols, which is the major storage form of lipids, is known as triglyceride biosynthesis.
It mainly occurs in the liver and adipose tissue, although it is also active in the intestine and other tissues to a lesser extent.
- Triglycerides provide fatty acids for membrane synthesis and signaling, act as energy stores, and shield cells from potentially harmful free lipids.
- This process includes activation of fatty acids to fatty acyl-CoA, making glycerol-3-phosphate, acylation to phosphatidic acid, conversion to diacylglycerol, and final acylation to form triacylglycerol.
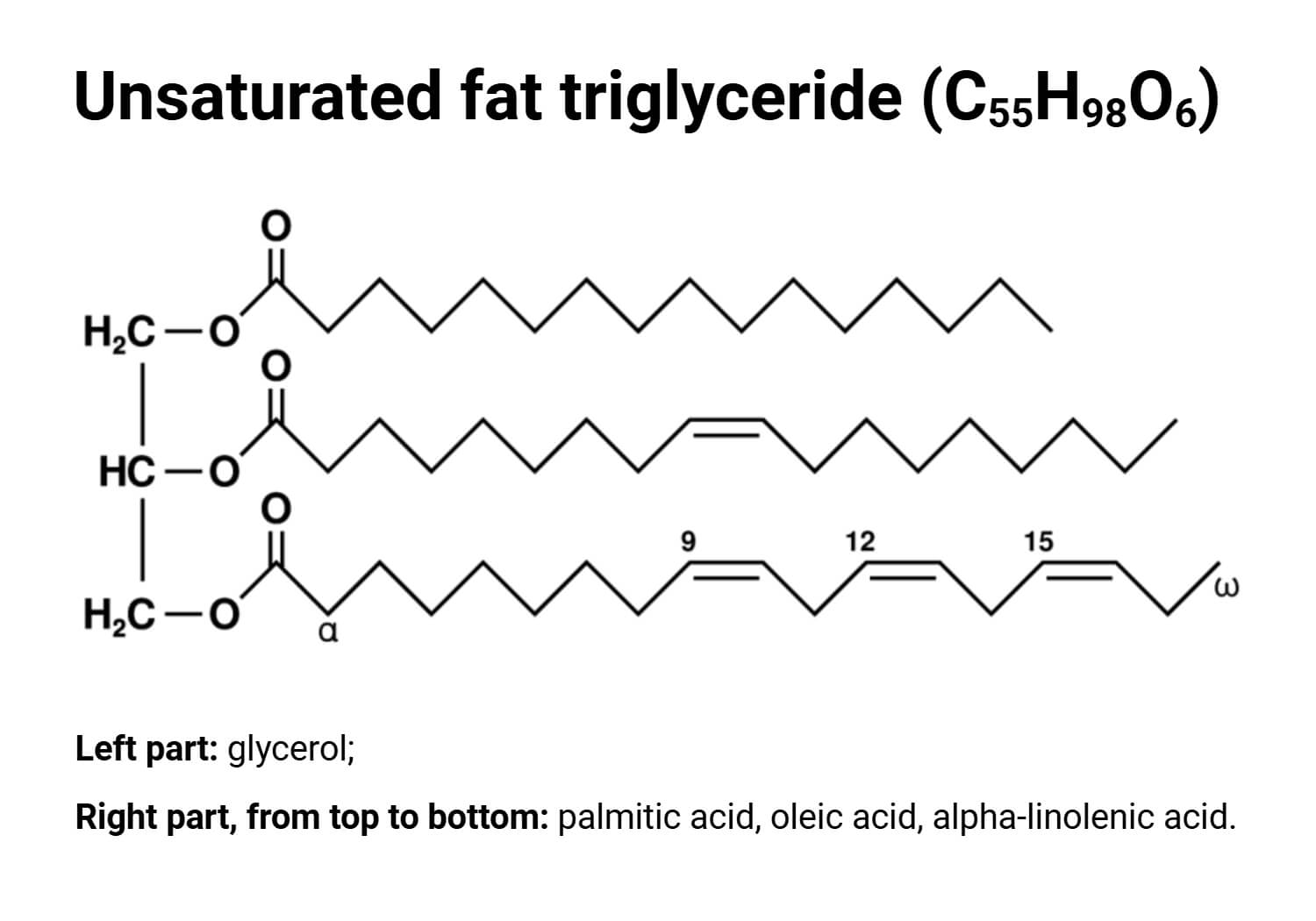
- The major synthesis of triglycerides occurs through the sn-glycerol-3-phosphate (Kennedy) pathway, with minor contributions by the dihydroxyacetone phosphate pathway and the monoacylglycerol pathway.
Site of Synthesis: Liver, Adipose Tissue, and Intestine
Liver
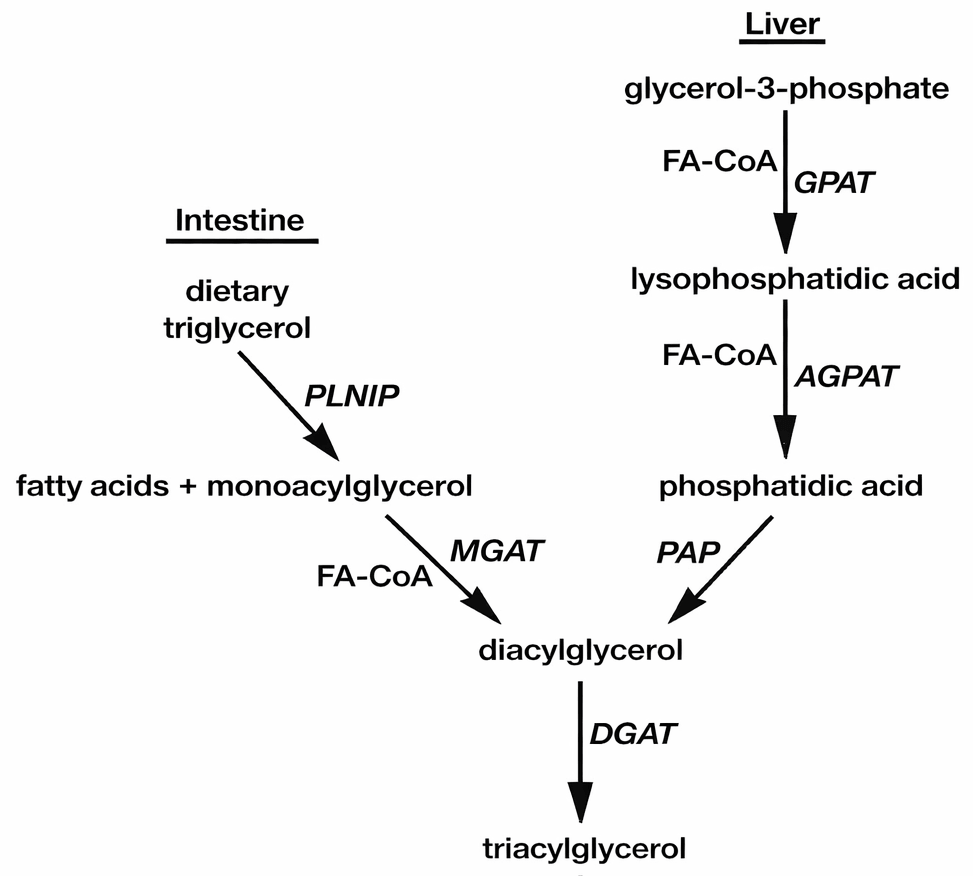
- Hepatocytes synthesize TGs by the sn-glycerol-3-phosphate (Kennedy) pathway, utilizing fatty acids (FA) and glycerol-3-phosphate.
- The final acylation is catalyzed by diacylglycerol acyltransferase 1,2 (DGAT1 and DGAT2), and DGAT2 TGs are secreted in very low-density lipoproteins (VLDL).
- Liver TG synthesis balances the uptake of FA, oxidation, and VLDL production; very little is stored in lipid droplets.
- The dysregulation will cause hepatic steatosis or non-alcoholic fatty liver disease.
Adipose Tissue
- TGs are produced by adipocytes as a long-term energy storage product from FA-coenzyme A (FA-CoA) and glycerol-3-phosphate.
- Preferably, the glycerol-3-phosphate pathway is the dominant one because adipocytes do not have glycerol kinase.
- Glycerol-3-phosphate acyltransferase (GPAT) and acylglycerophosphate acyltransferase (AGPAT) catalyze initial acylations; DGAT completes TG formation.
- TGs are stored in the lipid droplets of the cytoplasm and mobilized by lipolysis on demand for energy.
Intestine
- After luminal digestion, enterocytes resynthesize the dietary TGs using 2-monoacylglycerol (MAG) and FA.
- Monoacylglycerol acyltransferase (MGAT) is the major pathway that synthesizes diacylglycerol (DAG), followed by TG synthesized via DGAT.
- TGs are wrapped in apolipoprotein B48 in chylomicrons to be transported through the lymphatics.
- This is the process that allows the absorption of dietary fat and systemic lipid distribution.
Precursors Required: Sources of Glycerol-3-Phosphate and Fatty Acids
Sources of Glycerol-3-Phosphate
- The major precursor of the glycerol backbone in synthesizing TG is glycerol-3-phosphate (G3P), derived from two main sources.
- Glycerol kinase (GK) in the liver converts glycerol from dietary or circulating triglycerides into G3P.
- Glycerol-3-phosphate dehydrogenase produces glycerol-3-phosphate by reducing dihydroxyacetone phosphate (DHAP) from glycolysis in adipocytes and nonhepatic tissues.
Sources of fatty acids
- The chylomicron remnants carry the dietary triglyceride-derived fatty acids to the hepatocytes after re-esterification in the enterocytes.
- The free fatty acids released by adipose tissue under the influence of hormone-sensitive lipase are transported into the bloodstream attached to albumin and are sequestered in hepatic and adipose triglyceride production.
- The de novo lipogenesis in the liver transforms the surplus glucose and amino acids into fatty acids through the fatty acid synthase.
- Very low-density lipoprotein catabolism provides fatty acids that are reused in the production of triglycerides.
Activation of Fatty Acids: The Role of Fatty Acyl-CoA Synthetase
- The free fatty acids (FFA) are biologically inert and require conversion so that they can be utilized in the body to produce energy or to make lipids.
- This enzyme is present in the cytosol, outer mitochondrial membrane, and endoplasmic reticulum.
- Fatty acyl‑CoA synthetase (ACS) catalyzes ATP‑dependent formation of fatty acyl‑CoA, linking fatty acids with coenzyme A, producing AMP and PPi.
- During TG biosynthesis, fatty acyl-CoA is the activated fatty acid substrate, which is esterified onto glycerol-3-phosphate by acyltransferase enzymes to produce TG.
- This energy-demanding activation is necessary since free fatty acids are inert and cannot be used directly in biosynthesis.
Glycerol-3-Phosphate Pathway (Kennedy Pathway): Step-by-Step
- Triacylglycerols are synthesized from glycerol-3-phosphate and free fatty acids, both of which must be activated before initiating the biosynthetic pathway.
- Fatty acyl‑CoA synthetase (ACS) catalyzes ATP‑dependent formation of fatty acyl‑CoA, linking fatty acids with coenzyme A, producing AMP and PPi.
- Glycerol is converted to glycerol-3-phosphate by glycerol kinase in the liver using ATP; in adipose tissue, glycerol-3-phosphate is formed from dihydroxyacetone phosphate (DHAP) by glycerol-3-phosphate dehydrogenase, utilizing NADH.
- Glycerol-3-phosphate undergoes esterification with an activated fatty acyl-CoA at the 1-carbon position to form 1-acylglycerol-3-phosphate (lysophosphatidic acid) by the enzyme glycerol-3-phosphate acyltransferase (GPAT).
- The addition of one more fatty acyl-CoA to the 2nd carbon of Glycerol-3-phosphate produces 1,2-diacylglycerol-3-phosphate or phosphatidic acid by enzyme acylglycerol-3-phosphate acyltransferase (AGPAT).
- Phosphatidic acid is hydrolyzed by phosphatidic acid phosphohydrolase, releasing inorganic phosphate and forming diacylglycerol (DAG).
- Diacylglycerol (DAG) is again esterified with a third fatty acyl-CoA to form triacylglycerol, catalyzed by diacylglycerol acyltransferase (DGAT).
- DGAT is the rate-limiting step in the synthesis of triacylglycerol and phosphatidic acid, and DAG also serves as a precursor in phospholipid synthesis.
Key Enzymatic Steps: From GPAT to Diacylglycerol Acyltransferase (DGAT)
- Glycerol-3-phosphate acyltransferase (GPAT) adds a fatty acyl group to the sn-1 position of glycerol-3-phosphate and forms lysophosphatidic acid, and releases Coenzyme A (CoA).
- 1-Acylglycerol-3-phosphate acyltransferase (AGPAT) is an enzyme that attaches a second fatty acyl-CoA to form phosphatidic acid (1, 2-diacylglycerol-3-phosphate) with the release of CoA.
- Phosphatidic acid is broken down by phosphohydrolase, which takes in water and loses the inorganic phosphate (Pi) to produce diacylglycerol (DAG).
- Diacylglycerol acyltransferase (DGAT) adds a third fatty acyl-CoA to DAG, forming triacylglycerol (TAG) and releasing CoA.
The Monoacylglycerol Pathway: Specialized Synthesis in Enterocytes
- The monoacylglycerol (MAG) pathway is the most common postprandial triacylglycerol (TAG) resynthesis that occurs in small intestinal enterocytes, accounting for ~ 75–80 % of TAG formed after dietary fat absorption.
- In the lumen, pancreatic lipase hydrolyzes dietary TAGs into 2-monoacylglycerol (2-MAG) and free fatty acid (FFA), which enter enterocytes.
- Within the enterocyte, monoacylglycerol acyltransferase (MAGAT) enzymes acylate 2-MAG with fatty acyl-CoA, producing diacylglycerol (DAG), which is the precursor of TAG.
- This is followed by the addition of one more fatty acyl-CoA by the diacylglycerol acyltransferase (DGAT) to form TAG.
- TAGs that have been newly synthesized are coupled with phospholipids, cholesterol, and apoB-48 to create chylomicron, which is released into the lymph.
- In addition to being absorbed, 2-MAGs and FFAs are signaling molecules (GPR119, GPR40), which connect intestinal TAG metabolism to appetite, insulin secretion, and systemic energy balance.
Regulation of Lipogenesis: The Impact of Insulin and Glucagon
The impact of insulin
- Increases glucose uptake by the liver and adipose tissue.
- Activates two important lipogenic enzymes: fatty acid synthase (FAS) and acetyl-CoA carboxylase (ACC).
- Activates lipogenic gene transcription through SREBP-1 and other transcription factors.
- Promotes the synthesis of fatty acids and triglycerides.
Impact of glucagon
- Released when fasting or when blood sugar levels are low.
- Inhibits lipogenic enzymes and fatty acid synthesis.
- Inhibits lipogenic gene expression.
- Alters metabolism towards energy mobilization instead of energy storage.
Glucagon and insulin are antagonistic hormones that help to maintain energy homeostasis.
Transport and Storage: VLDL Formation and Lipid Droplets
VLDL formation
- In hepatocytes, VLDL is assembled to carry out endogenous triglycerides and cholesterol ester transport from the liver to the peripheral tissues.
- Microsomal triglyceride transfer protein transfers triglycerides to apolipoprotein B-100, resulting in the formation of nascent VLDL.
- In the blood, nascent VLDL picks up apo E and apo C-II from circulating HDL and becomes mature VLDL.
- Lipoprotein lipase (LPL) breaks down VLDL triglycerides in the capillaries of the peripheral tissues into glycerol and free fatty acids, which are diffused into the tissues.
- When the triglycerides are removed, VLDL is converted to a VLDL remnant, or intermediate-density lipoprotein (IDL).
- IDL undergoes hepatic uptake or conversion to LDL, retaining apo B-100 for further transport.
Lipid Droplets
- Lipid droplets (LDs) are intracellular organelles that contain neutral lipids (TAGs, cholesterol esters) in a hydrophobic core surrounded by a phospholipid monolayer and associated proteins.
- The processes of LD biogenesis start at the endoplasmic reticulum with the synthesis of neutral lipids, lens formation, the formation of the bud, growth, and maturation.
- When there is a surplus of energy, they store excess lipids and release fatty acids for β-oxidation through controlled lipolysis.
- LDs provide lipids to be used in membrane synthesis and the production of signaling molecules.
- Unregulated LD build-up has been associated with metabolic syndrome, obesity, insulin resistance, and non-alcoholic fatty liver disease (NAFLD).
Clinical Significance: Hypertriglyceridemia and Non-Alcoholic Fatty Liver Disease (NAFLD)
Hypertriglyceridemia
- Hypertriglyceridemia refers to the increased blood triglyceride (TG) levels (> 150 mg/dl) due to surplus fats in the blood.
- Etiology is multi-factorial: genetic disorders of lipid metabolism, excessive production of VLDL/chylomicron, defect in clearance of TRL, obesity, type 2 diabetes, hypothyroidism, medications, alcohol, and high-carbohydrate diets.
- Severe TG elevation (particularly >500mg/dl) is a significant risk factor for acute pancreatitis because TG hydrolysis causes the release of toxic free fatty acids that damage pancreatic tissue.
- Even mild-moderate hypertriglyceridemia is linked to atherosclerotic cardiovascular disease (ASCVD) and is considered an independent risk factor.
- High levels of triglycerides indicate excessive production and/or dysfunctional breakdown of VLDL and chylomicrons, resulting in pro-atherogenic remnant particles and inflammatory mediators.
Non-Alcoholic Fatty Liver Disease (NAFLD)
- Non-alcoholic fatty liver disease (NAFLD) is defined as the deposition of greater than 5% fat in the hepatocytes by histology in non-alcoholics.
- NAFLD is strongly related to metabolic dysfunction such as type 2 diabetes, insulin resistance, dyslipidemia, and metabolic syndrome characteristics.
- NAFLD is the leading chronic liver disease in the world, and it may advance into benign steatosis to NASH, fibrosis, cirrhosis, and hepatocellular carcinoma.
- It increases all-cause and cardiovascular morbidity and mortality and is an independent risk factor for cardiovascular disease (including coronary artery disease and hypertension).
- Pathogenesis includes oxidative stress, chronic inflammation, lipotoxicity, and insulin resistance-driven fat accumulation in hepatocytes, all of which contribute to fibrosis and progressive liver damage.
Conclusion
- Triglyceride biosynthesis is a key lipogenic process that produces triacylglycerols, the body’s primary lipid storage form, from fatty acids and glycerol.
- It usually occurs in the liver and adipose tissue, and the intestine is also important in resynthesizing triglycerides that are in the diet.
- The sn-glycerol-3-phosphate (Kennedy) is the main pathway, with the monoacylglycerol pathway being prevalent in enterocytes when food is consumed.
- Fatty acids should be activated to fatty acyl-CoA, and glycerol-3-phosphate is used as the backbone in the formation of triglycerides.
- Insulin stimulates the production of triglycerides and fatty acids, and glucagon suppresses the production of lipids, keeping energy homeostasis.
- The triglycerides are transported through VLDL and chylomicrons and stored in lipid droplets to be used as a source of energy.
- Hypertriglyceridemia, NAFLD, and metabolic disorders are caused by dysregulation of triglyceride synthesis, transport, or storage.
Reference
- Biology Insights. (n.d.). Lipid droplets: Function, structure, and role in disease. https://biologyinsights.com/lipid-droplets-function-structure-and-role-in-disease/
- Jakubowski, H., & Flatt, R. (n.d.). Biosynthesis of triacylglycerols. In Fundamentals of Biochemistry. LibreTexts.
- Jensen-Urstad, A. P., & Semenkovich, C. F. (2012). Fatty acid synthase and liver triglyceride metabolism: housekeeper or messenger?. Biochimica et biophysica acta, 1821(5), 747–753. https://doi.org/10.1016/j.bbalip.2011.09.017
- Kersten S. (2001). Mechanisms of nutritional and hormonal regulation of lipogenesis. EMBO reports, 2(4), 282–286. https://doi.org/10.1093/embo-reports/kve071
- Le, M. M., & Mohiuddin, S. S. (2023). Nonalcoholic fatty liver disease. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK587400/
- McFie, P. J., Patel, A., & Stone, S. J. (2022). The monoacylglycerol acyltransferase pathway contributes to triacylglycerol synthesis in HepG2 cells. Scientific reports, 12(1), 4943. https://doi.org/10.1038/s41598-022-08946-y
- McFie, P. J., Patel, A., & Stone, S. J. (2022). The monoacylglycerol acyltransferase pathway contributes to triacylglycerol synthesis in HepG2 cells. Scientific reports, 12(1), 4943. https://doi.org/10.1038/s41598-022-08946-y
- Ramakrishnan, N., & Auger, K. (2023). Hypertriglyceridemia. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK459368/
- Satyanarayana, U., & Chakrapani, U. (2013). Biochemistry (4th ed.). Elsevier.
- The Medical Biochemistry Page. (2025).Synthesis of triglycerides. https://themedicalbiochemistrypage.org/synthesis-of-triglycerides/
- Yaqub, S., Ananias, P., Shah, A., Luenam, K., Jose, A. M., Melo, J. P., Turkistani, A., & Mohammed, L. (2021). Decoding the Pathophysiology of Non-alcoholic Fatty Liver Disease Progressing to Non-alcoholic Steatohepatitis: A Systematic Review. Cureus, 13(9), e18201. https://doi.org/10.7759/cureus.18201
- Yen, C. L., Stone, S. J., Koliwad, S., Harris, C., & Farese, R. V., Jr (2008). Thematic review series: glycerolipids. DGAT enzymes and triacylglycerol biosynthesis. Journal of Lipid Research, 49(11), 2283–2301. https://doi.org/10.1194/jlr.R800018-JLR200
