Rheumatoid arthritis (RA) is a chronic inflammatory disorder that primarily targets the body’s joints, particularly those in the hands and feet.
- The condition is an autoimmune disease where the body’s immune system mistakenly attacks its own tissues, including the vital lining of the joints (synovium). This attack leads to painful swelling and prolonged inflammation. Over time, this chronic inflammation can result in bone erosion and joint deformity. In severe cases, RA can even affect and damage internal organs.
- While there is currently no cure for rheumatoid arthritis, early and consistent management is key to slowing its progression. Most cases are effectively managed through a combination of physiotherapy and a specific class of medications known as disease-modifying anti-rheumatic drugs (DMARDS)
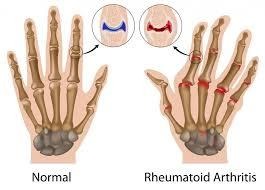
Understanding Rheumatoid Arthritis
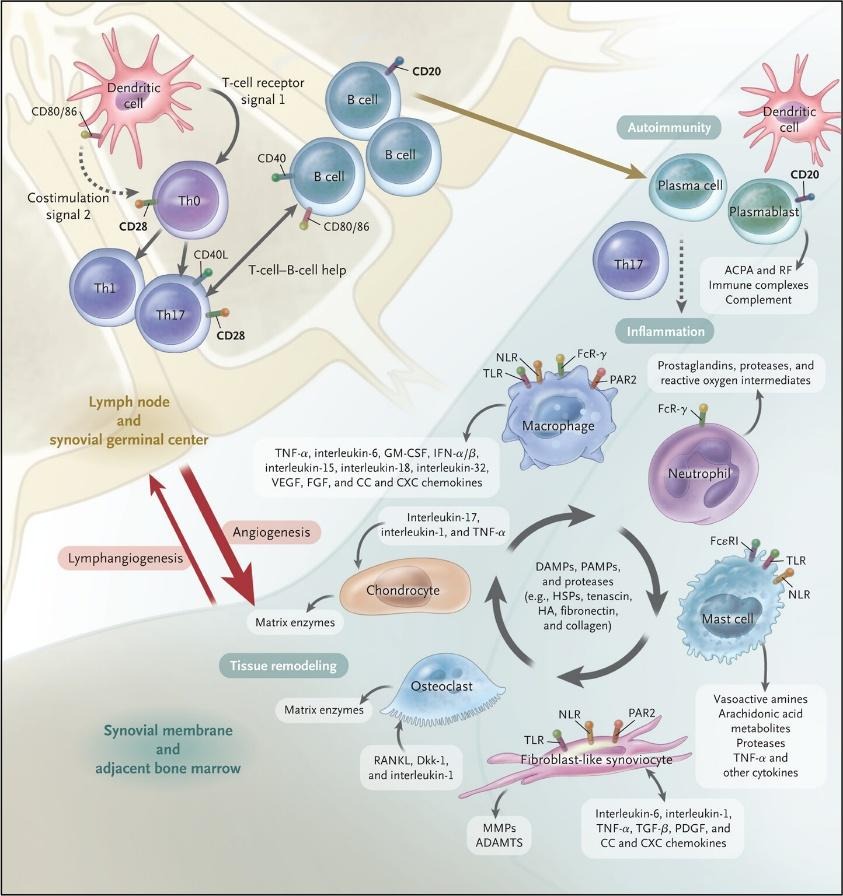
- The pathogenesis of RA is driven by a complex chain of events leading to joint inflammation and structural damage that primarily originates in the synovium (the joint lining). The synovial membrane is composed of various cells, notably Fibroblast-like Synoviocytes (FLS), which line the membrane. These cells are central to the pathogenesis of RA. The synovial membrane becomes inflamed (synovitis). While the exact trigger of the disorder remains unknown, several immune cells are involved in the initiation of this inflammation.
- Macrophages are present in the synovial membrane and act as key inflammatory regulators. They produce pro-inflammatory cytokines such as IL-1, IL-6, and TNF- alpha, which drive inflammation in the synovium. These cytokines powerfully stimulate and activate the FLS. This activation leads to the rapid proliferation of FLS. The FLS, along with these cytokines, assist in the expression of RANKL (Receptor Activator of Nuclear factor kappa B Ligand. It acts as the central regulator for osteoclasts which are cells that break down bone) stimulating osteoclast activity and consequently leading to bone erosion. Activated FLS also produce protease enzymes, which cause the breakdown of cartilage, initiating cartilage degradation. This destructive process is amplified by a feedback loop, as damaged cartilage also produces proteases. The ability of FLS to migrate from joint to joint is thought to contribute to the characteristic symmetrical arthritis seen in RA.
https://www.nejm.org/doi/full/10.1056/NEJMra1004965
- T-cells are another critical immune cell, making up to 50% of the immune infiltrate in the synovial membrane. They promote inflammation by secreting IL-17, which further stimulates macrophage activity and FLS proliferation cells also contribute to RANKL expression and bone destruction. Plasma cells are present in relatively lower numbers (approx. 5%) within the membrane, but they assist in inflammation through the localized production of cytokines and antibodies.
Events in the Synovial Fluid and Angiogenesis
- Neutrophils are found in the synovial fluid, producing proteases and Reactive Oxygen Species (ROS), which contribute to bone and cartilage degradation and inflammation.
- Immune Complexes are formed by the antibodies present in the fluid.
- Angiogenesis (new blood vessel formation) occurs, aiding the expression of adhesion molecules.
- Crucially, vascular permeability is increased, which facilitates the migration of immune cells from the synovium into the joint space.
Pre-RA Phase and Triggers
The preceding pre-rheumatoid arthritis phase involves systemic immune modifications:
Etiological Triggers
Genetics and Epigenetics: Having specific inherited gene variants, particularly those related to the HLA-DRB1 gene, significantly increases susceptibility to RA, with epigenetics influencing how these genes are expressed.
Smoking: Tobacco smoke is a powerful environmental trigger that is strongly associated with increasing the risk and severity of RA, especially in individuals with genetic predisposition.
The bacterium Porphyromonas gingivalis: This bacterium is hypothesized to trigger the autoantibody production (specifically Anti-CCP) in genetically susceptible individuals via a process called citrullination.
Systemic Autoantibody Production
Inflammation can cause the modification of autoantibodies (e.g., citrullination). These modified antigens are recognized by Antigen-Presenting Cells (APCs). The activated APCs initiate an immune response by migrating to the lymph nodes (the germinal centre). APCs activate CD4+ T-cells, which, in turn, stimulate B-cells via co -stimulation. B-cells proliferate, undergo class switching, become plasma cells, and produce autoantibodies. These cells then migrate to the joints throughout the body.
Two primary autoantibodies are responsible for RA pathology
Rheumatoid Factor (RF)
- RF is present in about 75% of RA cases.
- The main antibody of RF is the IgM antibody, which targets the Fc region (constant region) of the IgG Antibody.
- This interaction forms immune complexes IgM + IgG that promote inflammation.
Anti-Citrullinated Protein Antibody (ACPA)
- ACPA targets citrullinated proteins (like fibrin and filaggrin).
- The underlying mechanism is the conversion of the amino acid arginine residue into citrulline. The body mistakenly perceives the citrullinated protein as a foreign antigen and mounts an immune response against it.
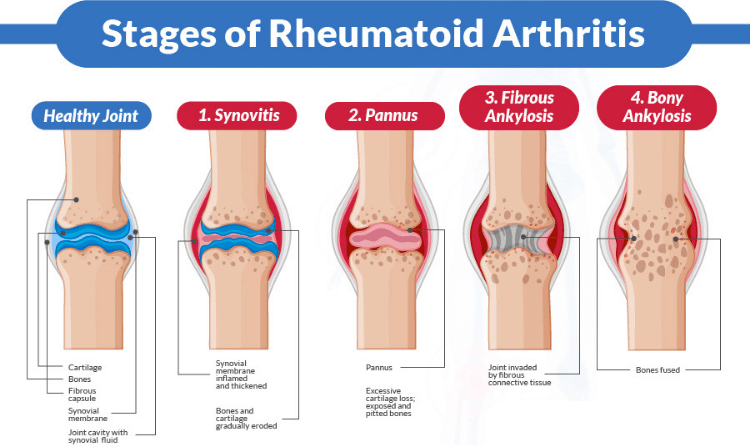
Stages of Rheumatoid arthritis
https://www.vaidam.com/knowledge-center/orthopaedic/everything-you-need-know-about-rheumatoid-arthritis
| Synovitis | Pannus Formation | Fibrous Ankylosis | Bony Ankylosis |
| Inflammatory cells infiltrate the synovial membrane, causing it to swell and thicken. Earliest symptoms: Joint pain, swelling, warmth, and morning stiffness. | The inflamed synovium grows into a highly aggressive mass (pannus) that releases destructive enzymes. The pannus directly invades and destroys the articular cartilage and underlying bone, leading to early erosions. | As cartilage and bone are destroyed, the gap between the joint surfaces is bridged by scar tissue (fibrous granulation tissue) The joint becomes stiff, fixed, and locked by this fibrous adhesion, resulting in significant loss of mobility and joint deformity. | The fibrous scar tissue connecting the joint surfaces eventually calcifies and ossifies (turns into solid bone). The joint is completely fused (immobilized). This is the irreversible, end-stage of joint destruction, leading to complete loss of function. |
Signs, Symptoms, and Impact of Rheumatoid Arthritis
Hallmark Symptoms:
The prominent sign of RA is symmetrical polyarthritis, which is the same joints getting affected on both the sides of the body e.g., both wrists or both hands. Morning stiffness is a key diagnostic feature, typically lasting for over an hour or following periods of inactivity.
Systemic Manifestations:
Rheumatoid Arthritis is not confined to the joints but is systemic, influencing the body as a whole. Patients often experience a general feeling of unwellness, known as profound fatigue, alongside possible low-grade fevers and unexplained weight loss. In approximately one-fifth to one-third of cases, RA can cause rheumatoid nodules which are firm, non-tender lumps that develop beneath the skin, typically over areas subjected to pressure, such as the back of the elbows.
Progression and Joint Damage:
Without timely intervention, chronic inflammation progresses to joint destruction. The hyper-proliferating pannus tissue actively breaks down the protective cartilage and underlying bone. This structural collapse causes permanent issues, including joint malformations and instability, which severely diminish a person’s ability to move and function independently. To protect the joints and preserve long-term mobility, early and assertive medical management is critical.
Diagnosis of Rheumatoid Arthritis
Diagnosing Rheumatoid Arthritis requires a multi-faceted approach, integrating symptoms, physical examination, specific blood tests, and imaging results. No single diagnostic test is definitive; instead, a healthcare provider establishes the diagnosis by recognizing a characteristic pattern across all these findings.
Laboratory Tests
Test kits are available in the markets for qualitative and quantitative detection of the RA specific antibodies.
In the diagnosis of Rheumatoid Arthritis, Anti-CCP antibodies stand out as the superior blood test. They are highly specific to RA and often appear very early, offering rheumatologists a crucial tool for timely diagnosis and effective treatment initiation. While Rheumatoid Factor (RF) is detected in most RA patients (about 80%), its value is constrained by its lower specificity, as it can also register positive results in people with other medical conditions or even in seemingly healthy individuals.
Inflammatory Markers
Blood tests like the Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) are employed to estimate the intensity of body-wide inflammation; elevated readings indicate high disease activity in RA. However, these markers are non-specific, as they rise in response to many conditions (infections, injuries). Therefore, their chief function is to monitor the aggressiveness of RA over time, not to serve as a standalone diagnostic confirmation.
Imaging: Mapping Structural Damage
Imaging techniques are crucial for visualizing the physical damage caused by RA and tracking its progression. While X-rays remain the standard for documenting chronic, advanced harm (such as bone loss/erosions and narrowed joint spaces), MRI and Ultrasound offer greater sensitivity. These advanced modalities allow physicians to detect early synovitis (joint lining inflammation) and subtle erosions far sooner than conventional X-rays, enabling more timely and effective therapeutic adjustments.
Management and Treatment of Rheumatoid Arthritis
- The overarching goal in treating Rheumatoid Arthritis (RA) is straightforward which is to achieve clinical remission or very low disease activity as quickly as possible. Early and aggressive treatment is absolutely critical because it is the only way to effectively halt the autoimmune process, prevent the irreversible joint damage, and ensure the patient retains maximum mobility and function over their lifetime.
- RA treatment utilizes a combination of medications designed to address both symptoms and the underlying disease mechanism.
- NSAIDs (Nonsteroidal Anti-inflammatory Drugs) and short-term Corticosteroids (like prednisone) fall under the Acute Symptom Relief drug category. These are fast-acting drugs used for immediate pain control and to reduce acute inflammation flares. They offer relief but do not change the course of the disease.
- Conventional DMARDs whose key example is Methotrexate is the foundational drug, often used in combination with others like sulfasalazine. It is Disease-Modifying Antirheumatic Drugs which are the backbone of RA therapy. They work gradually (over weeks to months) to suppress the immune system and slow the disease’s destructive progression.
- Biologics & Targeted Synthetics such as adalimumab, which targets TNF and Targeted Synthetic DMARDs (e.g., JAK inhibitors). These are advanced therapies reserved for cases where conventional DMARDs are insufficient. They operate by precisely targeting and blocking specific molecules (like TNF or JAK enzymes) within the inflammatory pathway.
Non-Pharmacological Strategies: While medication tackles the core disease, non-drug therapies are vital support tools used to manage symptoms, preserve physical function, and enhance quality of life in RA patients.
Physical Therapy (PT) focuses on maintaining flexibility, strength, and range of motion through targeted exercises and modalities (like heat/cold) whereas Occupational Therapy (OT) helps patients adapt daily routines by teaching joint protection methods and recommending assistive devices to minimize strain on damaged joints during essential activities.
Crucial adjustments include engaging in regular, low-impact exercise (e.g., swimming) to stabilize joints, ensuring it’s balanced with rest. A diet rich in omega-3s and antioxidants is advised for its general anti-inflammatory benefits. stress management (through methods like meditation) is important, as emotional stress can frequently trigger or intensify RA flares.
References
- Mueller, A.-L., Payandeh, Z., Mohammadkhani, N., Mubarak, S. M. H., Zakeri, A., Bahrami, A. A., Brockmueller, A., & Shakibaei, M. (2021). Recent advances in understanding the pathogenesis of rheumatoid arthritis: New treatment strategies. Cells, 10(11), 3017. https://doi.org/10.3390/cells10113017
- Castro-Sánchez, R., Roda-Navarro, P., & Al-Shahrour, F. (2017). Role of CD4+ T cells in rheumatoid arthritis. In R. T. B. J. R. T. P. A. A. R. E. C. A. C. & R. T. R. (Eds.), Physiology and Pathology of Autoimmune Diseases. IntechOpen. https://doi.org/10.5772/intechopen.72890
- Gao, S., [Other Authors]. (2024). Rheumatoid arthritis: Pathogenesis and therapeutic advances. MedComm, V(I), pages. https://doi.org/10.xxxx/xxxx
- Owen, J. A., Punt, J., & Waloop, S. A. (2013). Kuby Immunology (7th ed.). W. H. Freeman and Company.
- Rheumatoid Arthritis: The Continuum of Disease and Strategies for Prediction, Early Intervention, and Prevention | The Journal of Rheumatology
- Tadi, P., & Katta, A. (2018). Rheumatoid Arthritis: A Brief Overview of the Treatment. Medical Principles and Practice, 27(6), 501–507. https://doi.org/10.1159/000493390
- Long-term clinical outcomes in early rheumatoid arthritis that was treated-to-target in the BeSt and IMPROVED studies | Rheumatology | Oxford Academic
- https://www.youtube.com/watch?v=RqlBqtLKyNI
- Reitblat, T., Reitblat, O., & Elkayam, O. (2012). Unusual presenting syndrome of rheumatoid arthritis exacerbation. Open Journal of Rheumatology and Autoimmune Diseases, 2(1), 1–2. https://doi.org/10.4236/ojra.2012.21001
