Lipoproteins are complex biological particles composed of a core of triglycerides and cholesterol esters, surrounded by a shell of free cholesterol, phospholipids, and apolipoproteins.
- Lipoproteins are formed in the liver and intestine, arise from metabolic changes of precursor lipoproteins, or are assembled at cell membranes using lipids and apolipoproteins.
- They function as transport vehicles of lipids in blood plasma.
Structure of Lipoproteins
- The core of lipoprotein contains hydrophobic lipids such as triglycerides and cholesterol esters, which are protected from the aqueous environment.
- This core is surrounded by a monolayer of amphipathic molecules, consisting of phospholipids, free cholesterol, and apolipoproteins.
- Apolipoproteins contribute to both structural integrity and biological function.
Classification of Lipoproteins
Lipoproteins are a heterogeneous group, distinguished by differences in density and by the specific roles they perform in the circulatory system.
They can be classified into five main categories:
- Chylomicrons
- Produced in the intestinal mucosa, these are very low-density and large-sized particles.
- Mostly made of dietary triglycerides (approximately 84 %) and minimal protein.
- Its main role is to deliver exogenous triglycerides to the peripheral tissues.
- Very low-density lipoproteins (VLDL)
- Produced in the liver and enriched with triglycerides produced endogenously.
- It contains Apo B-100 as the structural apoprotein, along with Apo C and Apo E.
- Primarily involved in the transportation of endogenous triglycerides in the liver to tissues.
- Intermediate-density lipoproteins (IDL)
- This is formed as temporary particles when the VLDL is catabolized in the circulation.
- Exhibit intermediate density of mixed triglyceride and cholesterol.
- Act as a metabolic precursor of LDL or are taken up by the liver through Apo E-mediated receptors.
- Low-density lipoproteins (LDL)
- Formed as a result of additional metabolism of IDL.
- High in cholesterol and cholesterol ester, Apo B-100 is the only apoprotein.
- The key role includes transporting cholesterol to the peripheral tissues.
- High-density lipoproteins (HDL)
- Synthesized in the liver and intestine, the smallest and densest lipoprotein class.
- Characterized by high protein content (30–60%), mainly Apo A-I.
- Central role in reverse cholesterol transport, returning excess cholesterol to the liver.
The Role of Apoproteins: Functions and Types (ApoA, ApoB, ApoE)
- Apoproteins, also known as apolipoproteins, are the protein components of lipoproteins that bind with lipids such as cholesterol, triglycerides, and phospholipids.
- They stabilize lipoprotein structure, and this enables the movement of lipids in the blood.
- They are essential for the transport of hydrophobic lipids in the aqueous environment of the circulatory system.
- Apoproteins act as enzyme activators, receptor ligands, and play a role in lipoprotein metabolism and clearance.
Key Apoprotein Types
- ApoA (Apolipoprotein A)
- The main protein component of HDL is synthesized within the liver (70%) and intestine (30%).
- Promotes the removal of extra cholesterol from peripheral tissues and returns it to the liver for excretion by driving reverse cholesterol transport.
- Cardiovascular protection is linked to elevated ApoA levels.
- Apolipoprotein B (ApoB)
- Structural protein of VLDL, IDL, and LDL; present as ApoB-100 in liver-derived particles and ApoB-48 in intestinal chylomicrons.
- Vital for ligand binding to LDL receptors and lipoprotein assembly and secretion, which facilitates the delivery of cholesterol to cells.
- A higher number of atherogenic lipoprotein particles and a higher risk of cardiovascular disease are indicated by elevated ApoB.
- Apolipoprotein E (ApoE)
- present on HDL, VLDL, and chylomicron remnants.
- Act as a ligand for lipoprotein receptors, especially in the liver, facilitating clearance of remnant particles from the circulation.
- Additionally, ApoE plays a role in lipid transport in tissues other than the liver, such as the brain.
Exogenous Pathway: Metabolism of Chylomicrons from Diet
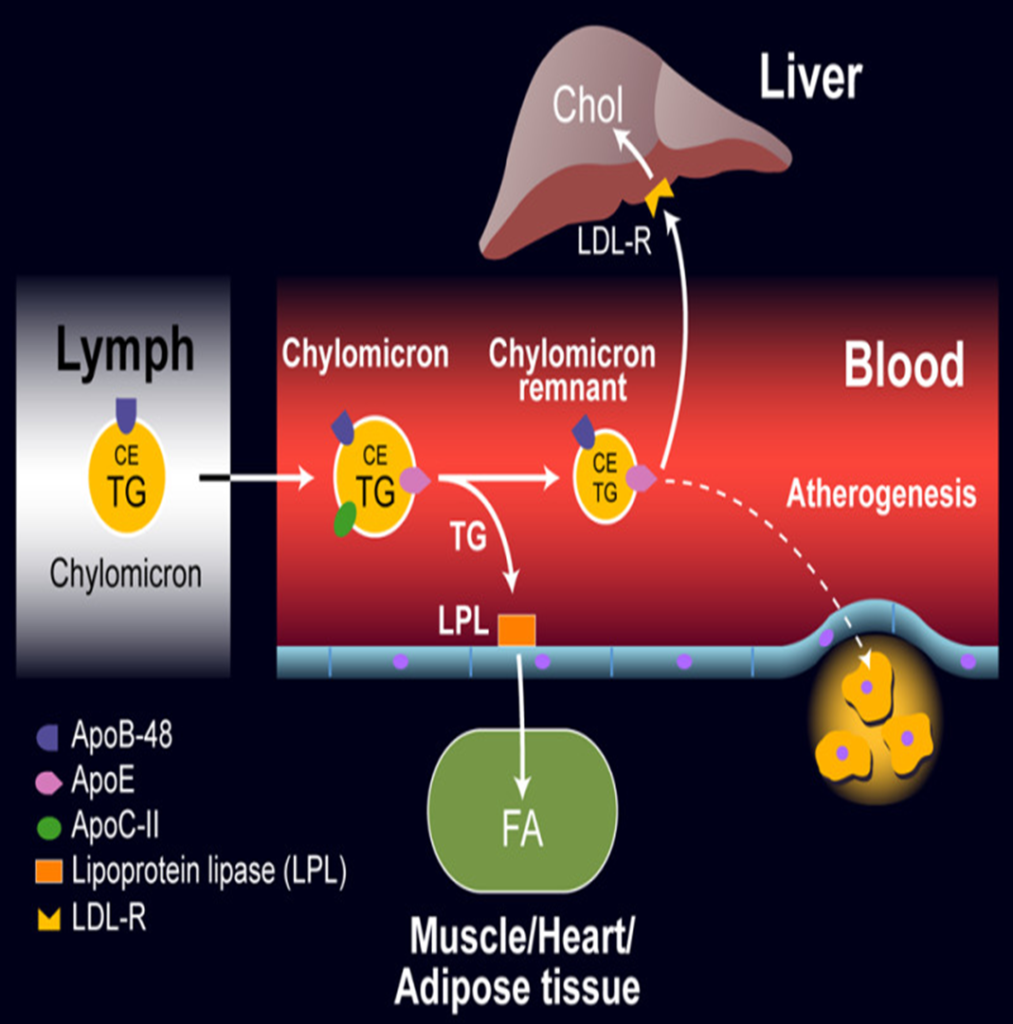
Formation of Chylomicrons
- In the intestinal lumen, pancreatic lipase breaks down dietary triglycerides (TG) and cholesterol esters into free fatty acids, monoacylglycerols, and glycerol.
- These elements in enterocytes are resynthesized into cholesterol esters and triglycerides.
- The nascent chylomicrons are assembled with triglycerides, cholesterol esters, phospholipids, and apolipoproteins (apo A and apo B-48).
Secretion into Lymph and Blood
- The nascent chylomicrons are discharged into the lymphatic system through the lacteals to be introduced into the bloodstream through the thoracic duct.
- Nascent CM picks up apo E, apo C-II from HDL.
Triglyceride Hydrolysis by Lipoprotein Lipase
- The Apo C-II stimulates lipoprotein lipase (LPL) in the endothelium of the capillaries.
- Triglycerides in chylomicrons are hydrolyzed by LPL, releasing fatty acids that are absorbed by peripheral tissues.
Formation of Chylomicron Remnants
- After most of the TG is removed, chylomicrons become smaller and are called chylomicron remnants.
- During the process, CM gives some of the phospholipids and apo C and apo A to HDL.
Hepatic uptake and processing
- Chylomicron remnants are endocytosed by binding to liver receptors (LDL receptor or LRP) through apo E.
- They bring dietary cholesterol, triglycerides, and HDL-derived cholesterol to the liver to be stored, to form bile salt, or to be excreted.
- The half-life of CM is short, less than 1 hour.
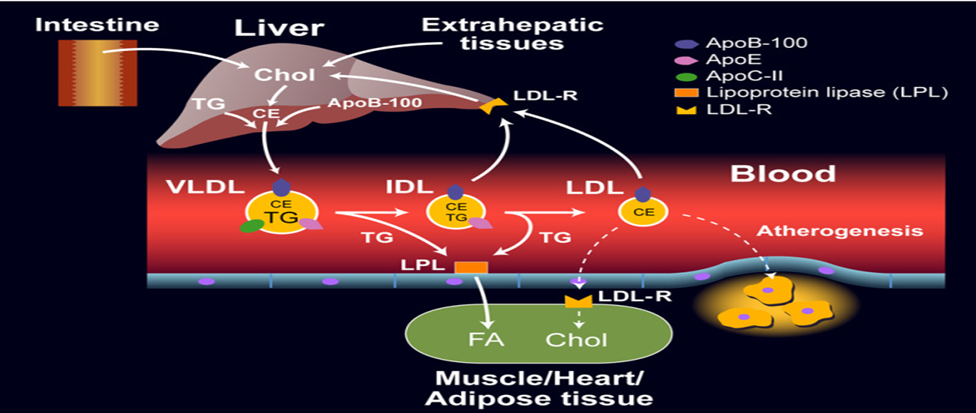
Endogenous Pathway: VLDL Synthesis and Transformation to LDL
- VLDL is made by the liver. The hepatocytes actively synthesize triglycerides (TG) out of fatty acids and glycerol.
- Hepatic cholesterol can either be derived from chylomicron remnants via the exogenous pathway or be synthesized on its own from cholesterol by hepatocytes when dietary cholesterol is insufficient.
- The triglycerides and cholesterol that are formed are packaged together in the liver and released into the circulation as nascent VLDL.
- The VLDL core is made up of triglycerides as the major lipid constituent.
- Nascent VLDL contains apo B-100.
- In the blood, nascent VLDL pick up apo E and apo C-II from circulating HDL and become mature VLDL.
- Lipoprotein lipase (LPL) breaks down VLDL triglycerides in the capillaries of the peripheral tissues into glycerol and free fatty acids, which are diffused into the tissues.
- LPL requires Apo C-II in its activation.
- When the triglycerides are removed, VLDL is converted to a VLDL remnant, or intermediate-density lipoprotein (IDL). In the process, the apo C-II is reintroduced to HDL.
- VLDL and HDL exchange lipids, with triglycerides being transferred to HDL and cholesteryl esters being transferred to VLDL, a process mediated by CETP.
- VLDL levels are enriched with cholesteryl esters.
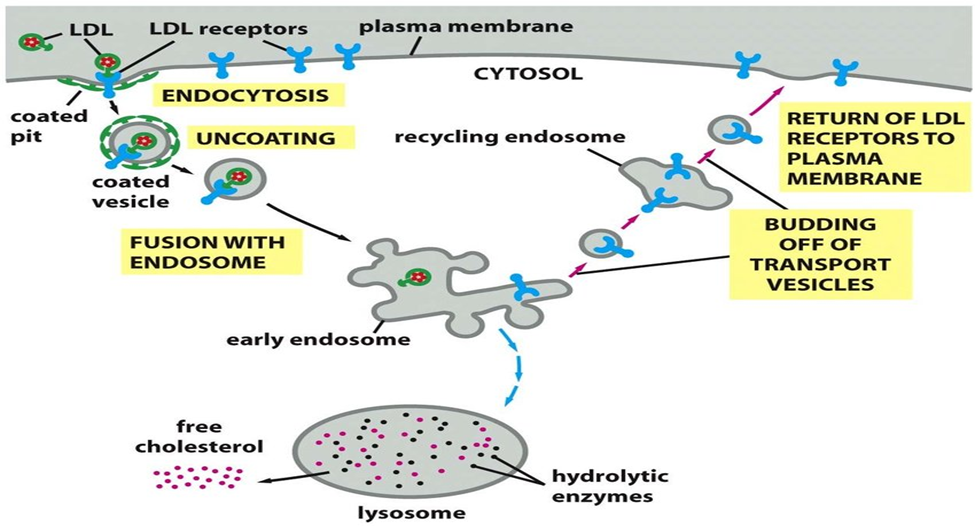
Low-Density Lipoprotein (LDL): Receptor-Mediated Endocytosis
- The process of LDL removal in the blood is regulated and is mainly done by hepatic cells and other peripheral tissue cells.
- LDL particles bind with certain LDL receptors, which are found on the cell surface.
- These LDL-receptor complexes then gather in specialized regions of the cell membrane called clathrin-coated pits.
- Such pits invert inwards and constrict to create clathrin-coated vesicles.
- Immediately after internalization, the clathrin coat disassembles from the vesicle and fuses with an early endosome.
- LDL is released from the receptor within the acidic environment of the endosome, and the receptor is returned to the plasma membrane.
- The LDL particles are then moved out of the endosome into the lysosome.
- Lysosomes break down the LDL proteins into amino acids, and the cholesterol esters are broken down into free cholesterol and fatty acids.
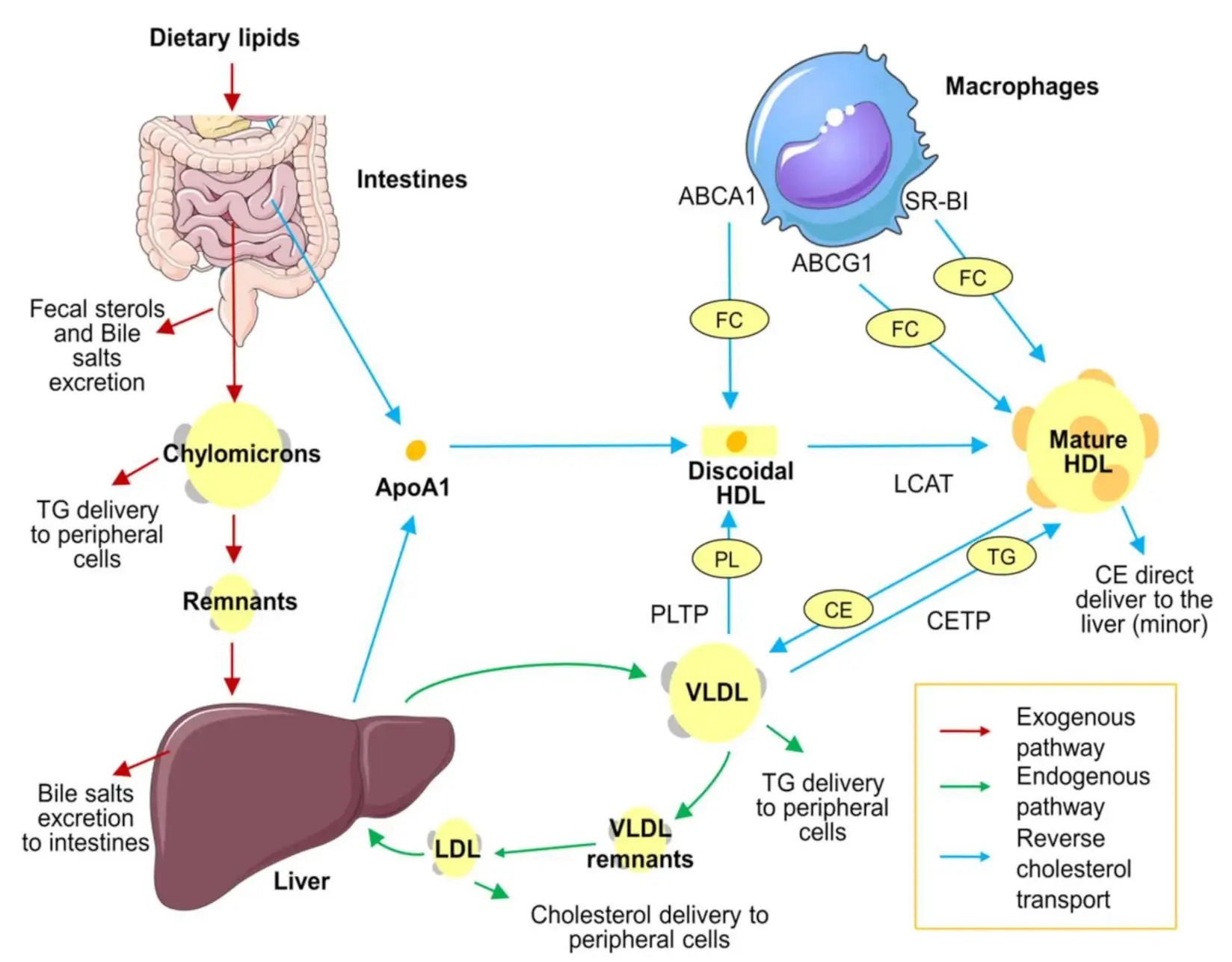
Reverse Cholesterol Transport: The Protective Role of HDL
- Reverse cholesterol transport refers to cellular transport of cholesterol from peripheral tissue to the liver, where it is excreted in feces as bile acids, cholesterol, and other catabolic products.
- HDL particles are formed in the blood by the addition of lipid to apo A-1, an apolipoprotein made by the liver and intestine and secreted into the blood.
- Approximately 70 percent of HDL apoproteins are Apo A-I, with Apo C-II and Apo E also present.
- Nascent HDL are disk-shaped, containing a high amount of phospholipids, principally phosphatidylcholine, and little cholesterol esters and triglycerides.
- Nascent HDL takes up cholesterol in chylomicrons, VLDL, and peripheral tissue in plasma through ATP-binding cassette protein-1 (ABCA1)
- Lecithin-cholesterol acyltransferase (LCAT) esterifies the obtained cholesterol immediately into cholesteryl esters of hydrophobic nature, which are transported into the HDL core.
- This is where nascent HDL is transformed into spherical mature HDL.
- HDL transports the cholesterol to the liver either directly through the SR-B1 receptor or indirectly through the transfer to the triglyceride-rich lipoproteins that are removed by the hepatic LDL receptors.
- In the liver, cholesterol is converted to bile acids or excreted into bile, completing elimination from the body.
Enzymatic Regulation: Lipoprotein Lipase (LPL) and LCAT
Lipoprotein Lipase (LPL):
- LPL breaks triglycerides (TG) of VLDL and chylomicrons into free fatty acids and glycerol.
- ApoC-II activates LPL, allowing the breakdown of triglycerides, whereas ApoC-III suppresses this effect.
- Insulin increases the LPL activity in the adipose tissue following meals; fasting or low insulin stimulates the LPL activity in muscle to provide energy.
- Repressed by ANGPTL4, which temporarily lowers lipid metabolism by destabilizing the active site of LPL
Lecithin–Cholesterol Acyltransferase (LCAT)
- LCAT is a plasma enzyme secreted by the liver, which esterifies free cholesterol to cholesteryl esters on HDL, leading to HDL maturation and reverse cholesterol transport.
- Activated by apo A-I on the HDL particles, which increases the esterification activity of LCAT.
- The control of hormones is indirect; sex hormones can affect the activity, but no hormone regulates LCAT directly, as LPL is regulated by insulin.
Lipid Transport Disorders: Dyslipidemia and Hypercholesterolemia
Dyslipidemia
- Dyslipidemia is a metabolic disorder characterized by abnormal quantities and patterns of lipids and lipoproteins in the blood.
- Typically manifested as Elevated LDL cholesterol, total cholesterol, triglycerides, and low HDL cholesterol concentrations.
- It is a significant risk factor for atherosclerosis and heart diseases such as heart attacks and strokes.
- There are primary causes (genetic predisposition) and secondary (lifestyle/medical) ones, including obesity, diabetes, unhealthy diet, and exercise.
Hypercholesterolemia
- Hypercholesterolemia is a lipid disorder that is characterized by excessively high levels of low-density lipoprotein cholesterol (LDL-C) in the blood, increasing the risk of atherosclerotic cardiovascular disease.
- Major risk factors include sedentary lifestyle, saturated/trans-fat diets, obesity, diabetes, and genetic predisposition (e.g., familial hypercholesterolemia).
- A condition usually asymptomatic up to the point of advanced atherosclerosis that leads to the occurrence of events like myocardial infarction or stroke.
- The management aims at lifestyle change (diet, exercise, quitting smoking) and pharmacotherapy using evidence-based statins and other lipid-lowering drugs to decrease LDL-C and cardiovascular risk.
Atherosclerosis: The Link Between Oxidized LDL and Plaque Formation
- Atherosclerosis is a condition, which is characterized by the deposition of lipids, fibrous matter, and calcification in the large arteries.
- Endothelial dysfunction initiates the process of atherosclerosis, which permits the entry of LDL into the arterial intima. Modified LDL, especially oxidized LDL (oxLDL), plays a central role.
- Because oxidized LDL avoids normal proteoglycan retention, it is more vulnerable to unregulated uptake by macrophage scavenger receptors.
- After being internalized, oxidized LDL causes the macrophages to express pro-inflammatory molecules, which also leads to chronic inflammation in the vessel wall.
- The macrophages that ingest large volumes of oxidized LDL develop into foam cells, forming the initial fatty streaks in atherosclerotic plaque.
- Further lipid deposition, inflammation, and recruitment of cells cause the further development of the plaque, which constricts the arteries and increases the risk of cardiovascular disease.
Clinical Assessment: Understanding Lipid Profile Tests
- A Lipid Profile Test, commonly referred to as a lipid panel, is a vital diagnostic test that measures the amount of the various fats or lipids in your blood.
- It quantifies the total cholesterol, LDL (bad) cholesterol, HDL (good) cholesterol, and triglycerides, which give an understanding of atherosclerotic risk and management.
Lipid Profile Diagnostic Criteria (mg/dL):
| Parameter | Desirable / Optimal | Borderline | High Risk |
|---|---|---|---|
| Total Cholesterol | <200 | 200–239 | ≥240 |
| LDL Cholesterol | <100 | 130–159 | ≥160 |
| HDL Cholesterol | ≥60 | 40–59 | <40 |
| Triglycerides | <150 | 150–199 | ≥200 |
Conclusion
- Lipoproteins are particles produced in the liver and intestines and carry triglycerides and cholesterol in the blood.
- They contain a hydrophobic core and an apolipoprotein on the surface; they are chylomicrons, VLDL, IDL, LDL, and HDL.
- The ApoA facilitates reverse cholesterol transport in HDL, ApoB facilitates VLDL/LDL structure and receptor binding, and ApoE clears remnants.
- Dietary lipids are transported through chylomicron (exogenous), liver lipids through VLDL-LDL (endogenous).
- LDL transports cholesterol to the cell; HDL cleanses the excess cholesterol.
- LPL and LCAT control the hydrolysis of lipids and the maturation of HDL.
- Lipid profile tests measure dyslipidemia and hypercholesterolemia, which encourage atherosclerosis through oxidized LDL.
Reference
- Alberts, B., Johnson, A., Lewis, J., Raff, M., Roberts, K., & Walter, P. (2008). Molecular biology of the cell (5th ed.). Garland Science.
- Apollo Hospitals. (n.d.). Lipid Profile Test – Purpose, Results, Normal Range.
- Biology Insights. (n.d.). Lipoproteins: Structure, types, and their role in lipid metabolism.
- Biology Insights. (n.d.). LPL biochemistry: Mechanism, regulation, and health.
- BMJ Best Practice. (2025). Hypercholesterolemia (topic overview). https://bestpractice.bmj.com/topics/en-gb/170
- DoveMed. (n.d.). Biochemistry of chylomicron: Structure, metabolism, and functions.
- Feingold, K. R. (2024). Introduction to lipids and lipoproteins. In Endotext. MDText.com, Inc. https://www.ncbi.nlm.nih.gov/books/NBK305896/
- Hajera, L., Reddy, M. C., & Reddy, K. J. (2021). A review of lipoproteins… World Journal of Pharmaceutical Research, 10(14), 341-356.
- Jebari-Benslaiman, S., Galicia-García, U., Larrea-Sebal, A., Olaetxea, J. R., Alloza, I., Vandenbroeck, K., Benito-Vicente, A., & Martín, C. (2022). Pathophysiology of Atherosclerosis. International Journal of Molecular Sciences, 23(6), 3346. https://doi.org/10.3390/ijms23063346
- Lab Tests Guide. (n.d.). Apoprotein. https://www.labtestsguide.com/apoprotein
- LIPID MAPS LipidWeb. (n.d.). Plasma lipoproteins.
- Merck Manuals Consumer Version. (n.d.). Dyslipidemia: Treatment.
- Øie, C. I. (2010). New insights into the clearance of tissue factor pathway inhibitor (TFPI) and unfractionated heparin (UFH) [Doctoral dissertation, University of Tromsø]. Munin.
- Ouimet, M., Barrett, T. J., & Fisher, E. A. (2019). HDL and Reverse Cholesterol Transport. Circulation research, 124(10), 1505–1518. https://doi.org/10.1161/CIRCRESAHA.119.312617
