- Ketone body metabolism is a metabolic pathway in which the body adapts to use fats as a primary energy source.
- The synthesis of ketone bodies occurs in the liver when the amount of acetyl-CoA exceeds the oxidative capacity of the TCA cycle.
- The enzymes for ketone body synthesis are in the mitochondrial matrix.
- Acetyl CoA, formed by oxidation of fatty acids, pyruvate, or some amino acids, is the precursor for ketone bodies.
- The metabolism of ketone bodies converts hepatic acetyl-CoA into acetoacetate and 2-hydroxybutyrate to supply energy to extrahepatic tissues.
- HMG-CoA synthase 2 catalyzes the rate-limiting step to generate HMG-CoA, and HMG-CoA lyase generates acetoacetate; acetoacetate is reduced to ϐ-hydroxybutyrate.
- Ketone bodies are exported and absorbed by extrahepatic tissues through monocarboxylate transporters (MCTs).
- In extrahepatic mitochondria, BDH1, succinyl-CoA:3-oxoacid CoA transferase (SCOT), and ACAT1 convert ketone bodies to acetyl-CoA, which is used in the TCA cycle to generate ATP.
What are Ketone Bodies?
Ketone bodies are water-soluble compounds with ketone groups that are synthesized in the liver using fatty acids in a process known as ketogenesis.
- Ketone bodies are produced in a caloric restriction condition, e.g., fasting, carbohydrate-restricted diets, starvation, intense exercise, alcoholism, or untreated type 1 diabetes.
- The ketone bodies can supply 50-70% of the energy requirements of the brain.
- The compounds, namely acetone, aceto-acetate, and β-hydroxybutyrate (or 3-hydroxybutyrate), are known as ketone bodies.
Acetoacetate (AcAc)
- The first ketone body formed in ketogenesis from two acetyl-CoA molecules via β-oxidation and the HMG-CoA intermediate in liver mitochondria.
- It serves as the precursor for the other ketones, such as β-hydroxybutyrate and acetone.
β -hydroxybutyrate
- These are produced by reducing acetoacetate with the help of β-hydroxybutyrate dehydrogenase.
- It is the most abundant ketone body that constitutes approximately 70 % of the total ketone bodies.
- A precursor of energy to the brain and muscles, it is also an important signaling molecule that can regulate the expression of genes, inflammation, and oxidative stress.
Acetone
- This is formed primarily by spontaneous (non-enzymatic) decarboxylation of acetoacetate.
- It is the least abundant ketone body, which is volatile and is primarily lost through breath or urine and not utilized as energy.
- Inefficiently converted back to acetyl-CoA and is not used much by metabolism as AcAc and BHB.
Ketogenesis: The Pathway of Synthesis in Liver Mitochondria
- Ketogenesis takes place solely in the mitochondrial matrix of hepatocytes.
- Acetyl-CoA is the main precursor, which is produced mainly by the process of fatty acid β-oxidation, and from pyruvate and ketogenic amino acids (e.g., leucine, lysine, phenylalanine).
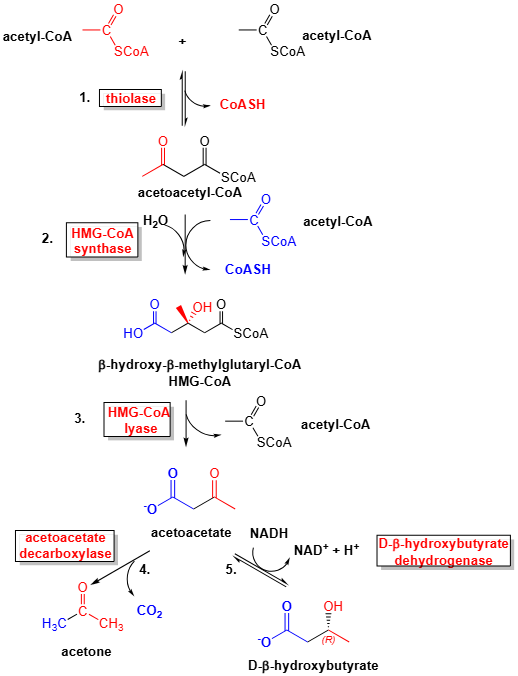
Step 1: Formation of Acetoacetyl-CoA
- Two molecules of Acetyl-CoA undergo condensation to form acetoacetyl-CoA, which is catalyzed by thiolase.
- This reaction is the opposite of the last step of ϐ-oxidation, and it connects fatty acid oxidation with ketone body production.
Step 2: Formation of HMG-CoA
- The acetoacetyl-CoA is then reacted with another acetyl-CoA to produce 2-hydroxy-2-methylglutaryl-CoA (HMG-CoA).
- This step is catalyzed by the rate-limiting and regulatory enzyme of ketogenesis, HMG-CoA synthase.
Step 3: Formation of Acetoacetate
- The HMG-CoA is cleaved by HMG-CoA lyase to produce acetoacetate, which is the first ketone body, with the release of one acetyl-CoA.
Step 4: Formation of Acetone
- Some of the acetoacetate spontaneously decarboxylates to acetone, a volatile ketone body which is mostly excreted.
Step 5: Formation of β-Hydroxybutyrate
- β-Hydroxybutyrate dehydrogenase catalyzes the reversible reduction of acetoacetate to β-hydroxybutyrate using NADH as an electron donor, reflecting the mitochondrial redox state.
Key Enzymes in Synthesis: The Role of HMG-CoA Synthase
- HMG-CoA synthase catalyzes the condensation of acetyl-CoA with acetoacetyl-CoA in liver mitochondria, producing 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA).
- The condensation of acetyl-CoA with acetoacetyl-CoA in liver mitochondria produces 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA) with the help of HMG-CoA synthase.
- It is the rate-limiting and committed step of the ketone body synthesis.
- The enzyme operates via a BI-substrate ping-pong mechanism: the acetyl group binds to Cys111, forming an acetyl-enzyme intermediate, then condenses with acetoacetyl-CoA via an enolate intermediate.
- Starvation, low insulin, and a high-fat diet stimulate activity.
- This step connects acetyl-CoA from β oxidation to the generation of ketone bodies and production of energy during fasting.
Ketolysis: How Peripheral Tissues Utilize Ketone Bodies for Fuel
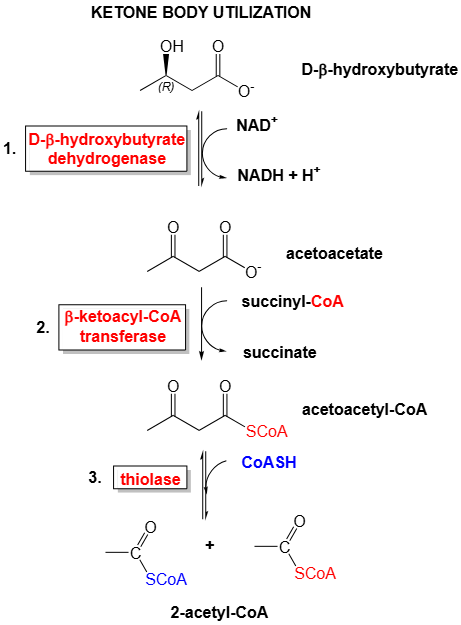
- Ketone bodies ( ϐ-hydroxybutyrate and acetoacetate) are synthesized in the liver and transported to the peripheral tissues (brain, heart, muscle, and kidney) through monocarboxylate transporters (MCTs).
- β-hydroxybutyrate is oxidized to acetoacetate in extrahepatic mitochondria by β-hydroxybutyrate dehydrogenase (BDH1).
- Succinyl-CoA:3-oxoacid CoA transferase (SCOT/OXCT1) transfers CoA from succinyl-CoA to acetoacetate, forming acetoacetyl-CoA — the rate-limiting step of ketolysis, absent in the liver to prevent futile cycling.
- Acetoacetyl-CoA is then cleaved by acetoacetyl-CoA thiolase (ACAT1) to yield two acetyl-CoA molecules.
- The acetyl-CoA enters the TCA cycle, and the electron transport chain yields ATP and supplies significant energy when glucose is in short supply.
Why the Liver Cannot Utilize Ketone Bodies: The Missing Enzyme (Thiophorase)
- Ketone bodies (AcAc and β-hydroxybutyrate) are synthesized in liver mitochondria but cannot be used there for energy.
- Hepatocytes do not have succinyl-CoA: acetoacetate CoA transferase (SCOT/thiophorase), which is the principal enzyme that breaks ketone bodies down to acetoacetyl-CoA to enter the TCA cycle.
- The lack of SCOT in the liver will ensure that there is no futile consumption of the ketone bodies produced by the liver, as they are exported to be used in the periphery.
Regulation of Ketogenesis: The Balance of Insulin and Glucagon
- Ketogenesis is a hormonally controlled process where insulin acts as the inhibitor and glucagon as the main stimulator of the ketogenesis process.
- Insulin has a potent effect on inhibiting the process of ketogenesis by inhibiting hormone-sensitive lipase, which diminishes the release of free fatty acids into the bloodstream.
- An increase in insulin levels suppresses fatty acid oxidation and the formation of ketone bodies in the liver.
- Reduced levels of insulin (fasting, starvation, uncontrolled diabetes) allow increased lipolysis and ketogenesis.
- Glucagon stimulates ketogenesis through the mobilization of fatty acids and increases the level of ketone production in the liver.
Physiological Ketosis vs. Diabetic Ketoacidosis (DKA): Clinical Differences
Physiological Ketosis
- occurs when blood glucose remains normal, but liver ketones increase due to low glucose availability (fasting, low-carb diet, exercise).
- There is no dangerous blood acidification; ketone levels are somewhat elevated but within acceptable limits.
- The body effectively uses ketones as a substitute fuel; insulin is present and working.
- There is no metabolic disturbance, and the blood pH stays normal.
- Generally helpful, intentional, and secure under supervision.
Diabetic Ketoacidosis (DKA)
- Outcomes of insulin insufficiency in diabetes, usually type 1, cause unregulated fat breakdown and extreme accumulation of ketones.
- Significantly high ketone (>2.9 mmol/L) and glucose (>13.8 mmol/L) concentrations in the blood.
- Blood acidifies (low pH), hyperglycemia, dehydration, and electrolyte imbalance develop.
- Potentially life-threatening; may result in coma or death without immediate medical intervention.
The Role of Ketone Bodies During Starvation and Prolonged Fasting
- Early fasting depletes glycogen stores and increases lipolysis, which produces acetyl-CoA from fatty acids that are β-oxidized in the liver. Excess acetyl-CoA is then transformed into ketone bodies.
- As starvation gets worse, the brain uses ketone bodies as a primary energy source, which lowers the need for glucose and prevents excessive proteolysis of muscle protein.
- The brain, heart, and skeletal muscle are increasingly powered by ketone bodies during the protein conservation phase, reducing dependency on gluconeogenesis from amino acids.
- Additionally, the increase in ketones has a negative feedback effect on muscle proteolysis, which helps to preserve body protein.
- When carbohydrate availability is low, ketone utilization maintains ATP production in critical tissues and promotes energy homeostasis.
ATP Yield from Ketone Body Oxidation
- β-hydroxybutyrate dehydrogenase first oxidizes β-hydroxybutyrate (BHB) to acetoacetate (AcAc), generating 1 NADH → 2.5 ATP.
- SCOT (succinyl-CoA:3-oxoacid CoA transferase) converts acetoacetate to acetoacetyl-CoA, which is subsequently divided into two acetyl-CoA molecules by thiolase.
- All acetyl-CoAs go into the TCA cycle, yielding:
- 3 NADH → 7.5 ATP
- 1 FADH₂ → 1.5 ATP
- 1 GTP → 1 ATP
- Total ~10 ATP per acetyl-CoA.
- Acetoacetate is broken down into two acetyl-CoA molecules, giving about 19 ATP after subtracting the 1 ATP needed for its activation.
- β-hydroxybutyrate is a slightly more efficient energy source because it first produces an extra NADH (about 2.5 ATP) before forming two acetyl-CoA. This results in a total of about 22.5 ATP.
Conclusion
- Ketone bodies (acetoacetate, β-hydroxybutyrate, acetone) are water-soluble fuels synthesized in liver mitochondria from acetyl-CoA when glucose is scarce.
- Low levels of insulin and high levels of glucagon control synthesis (ketogenesis), and the rate-limiting enzyme is the HMG-CoA synthase.
- Ketone bodies cannot be used by the liver because of the lack of enzyme SCOT (thiophorase) and are exported to peripheral tissues.
- Through the process of ketolysis, tissues such as the brain and heart produce approximately 22.5 ATP per β-hydroxybutyrate by converting ketones back to acetyl-CoA.
- Diabetic ketoacidosis is a dangerous pathological state of insulin deficiency, whereas physiological ketosis is an adaptive reaction to fasting.
- Overall, ketone metabolism preserves protein and glucose during starvation by offering an essential alternative energy source.
References
- Dhillon, K. K., & Gupta, S. (2023). Biochemistry, ketone metabolism. In StatPearls. StatPearls Publishing.
- Diabetes South Africa. (2022, November 30). Physiological ketosis vs diabetic ketoacidosis.
- Hwang, C. Y., Choe, W., Yoon, K. S., Ha, J., Kim, S. S., Yeo, E. J., & Kang, I. (2022). Molecular Mechanisms for Ketone Body Metabolism, Signaling Functions, and Therapeutic Potential in Cancer. Nutrients, 14(22), 4932. https://doi.org/10.3390/nu14224932
- Jakubowski, H., & Flatt, J. (n.d.). Ketone bodies. In Fundamentals of Biochemistry. LibreTexts.
- Laffel L. (1999). Ketone bodies: a review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes/metabolism research and reviews, 15(6), 412–426. https://doi.org/10.1002/(sici)1520-7560(199911/12)15:6<412::aid-dmrr72>3.0.co;2-8
- Puchalska, P., & Crawford, P. A. (2017). Multi-dimensional Roles of Ketone Bodies in Fuel Metabolism, Signaling, and Therapeutics. Cell metabolism, 25(2), 262–284. https://doi.org/10.1016/j.cmet.2016.12.022
- Rebello, C. J., Zhang, D., Anderson, J. C., Bowman, R. F., Peeke, P. M., & Greenway, F. L. (2025). From starvation to time-restricted eating: a review of fasting physiology. International journal of obesity (2005), 49(1), 43–48. https://doi.org/10.1038/s41366-024-01641-0
- Satyanarayana, U., & Chakrapani, U. (2013). Biochemistry (4th ed.). Elsevier.
