Enzymes are biological catalysts, primarily protein macromolecules, that act as highly efficient catalysts in living systems. Their fundamental role is to significantly accelerate (speed up) biochemical reactions without being used up themselves, making them vital for essential life processes like metabolism, digestion, and nerve signalling. They are the drivers of nearly all metabolic processes in an organism.
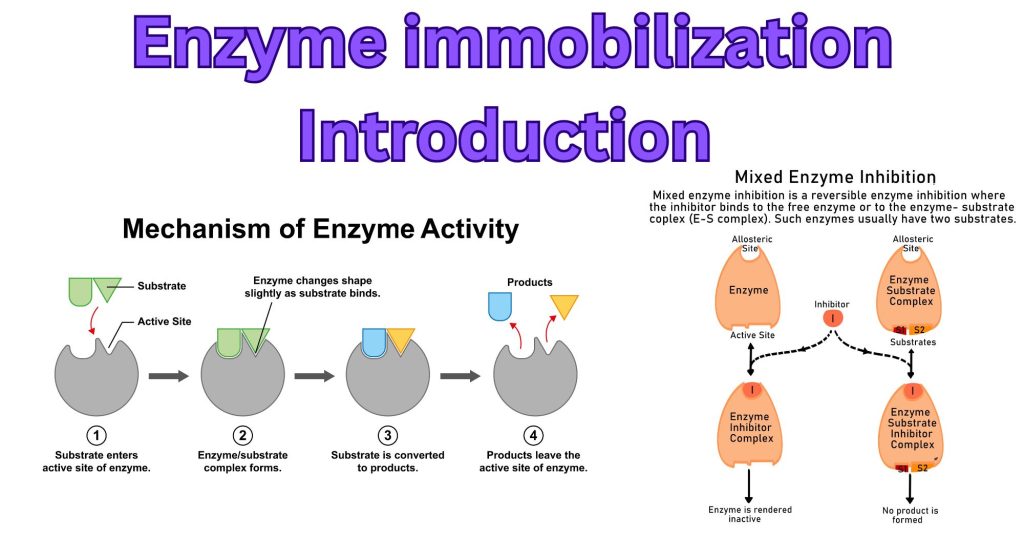
Enzymes possess a unique three-dimensional structure containing an active site. This site is designed to bind to a specific reactant, called the substrate, typically following an induced-fit model (a slight shape adjustment upon binding).
Enzymes have specific interactions, i.e., a specific substrate reacts with the active site, converting the substrate into the final product. This specific interaction means enzymes are highly specialized, often catalyzing only one or a few types of reactions
Enzymes possess extraordinary catalytic power, which allows biochemical reactions to occur at speeds necessary for life. They dramatically accelerate reaction rates, often making them proceed millions or even trillions of times faster than the uncatalyzed version.
The turnover rate (kcat), which is the maximum number of substrate molecules an enzyme can convert to product per second, is fundamentally determined by the efficiency of the active site and is influenced by several environmental and chemical factors
Initially, the rate is limited by how often the enzyme and substrate collide. However, the maximum turnover rate (Vmax) is reached when the enzyme becomes saturated, meaning all active sites are constantly occupied. At this point, adding more substrate won’t increase kcat, as the enzyme is working at its peak speed.
Increasing temperature generally increases the rate of molecular collisions and therefore the reaction rate, up to a point. Beyond the enzyme’s optimal temperature 37 C for human enzymes), the enzyme rapidly begins to denature (unfold), causing the turnover rate to plummet.
Enzymes have a narrow optimal pH range where their active site’s structure and the electrical charge of the substrate are perfectly aligned for catalysis.
The high cost of enzymes, especially those used in industry or research, presents a major practical challenge. Manufacturing requires intricate processes like fermentation or cultivation to grow the source organisms (usually microbes) that produce the enzyme. Getting the enzyme in a usable, high-purity state involves expensive and time-consuming steps like chromatography to separate it from other cellular materials. Enzymes’ inherent fragility necessitates special, costly measures (such as refrigeration, specific chemical buffers, or stabilizers) during both shipping and long-term storage to prevent denaturation and maintain their activity
Enzyme Immobilization: Definition, primary goal
Enzyme immobilization is the process of anchoring soluble enzyme molecules by either physically trapping or chemically bonding them to a stable, non-dissolving solid material (a support matrix). The goal is to lock the enzyme in place, restricting its movement to a specific area while fully preserving its ability to catalyse reactions. It effectively transforms a floating, liquid-based catalyst into a fixed, solid-based catalyst.
The fundamental reason for immobilizing enzymes is to cut down on operational costs in industrial and analytical settings by enabling the enzyme’s efficient, repeated, and continuous application.
Immobilization makes the expensive enzyme easily recoverable and separable from the products. This means the same enzyme can be utilized for numerous reaction cycles, eliminating the need to constantly buy new catalysts.
Anchoring the enzyme to a solid support often makes it much more resilient to damaging conditions (like heat, extreme pH, or solvents). This prevents the enzyme from unfolding (denaturation) and ensures it maintains its activity for a significantly longer period.
Because the enzyme is physically fixed to a solid, it doesn’t contaminate the final liquid product. This eliminates complex and costly steps needed to remove the enzyme from the solution during purification.
Immobilized enzymes are perfectly suited for continuous flow reactors. This enables automated, more predictable, and highly efficient industrial manufacturing processes.
Brief Historical Perspective of Enzyme Immobilization
The concept of enzyme immobilization, while seeing its major development in the mid-20th century, has a surprisingly early scientific root, but it was the economic pressures of industrial application that truly brought it to the forefront
The first scientific observation relevant to immobilization was made by Nelson and Griffin, who noted that the enzyme invertase retained its activity even after being adsorbed onto a solid material like charcoal. This demonstrated that an enzyme didn’t need to be soluble to function.
The technology crossed from the lab into the factory when Chibata and co-workers in Japan successfully immobilized the enzyme aminoacylase (from Aspergillus oryzae). This process was used on a commercial scale to produce L-amino acids, the first large-scale industrial use of an immobilized enzyme, marking the true dawn of the field.
The success of the aminoacylase process sparked widespread global research, cementing enzyme immobilization as a vital area of enzyme technology for food, pharmaceutical, and chemical industries.
Classification of the Enzyme Immobilization by Support Material
The strategic selection of a carrier material is the single most critical factor in enzyme immobilization, as it not only dictates the choice between physical and chemical methods but fundamentally determines the immobilized system’s enhanced thermal stability and long-term industrial applicability.
- Inorganic Carriers (Rigid and Stable):
Inorganic carriers provide a rigid and stable nature for enzymes requiring high physical resistance in harsh conditions. Silica and porous glass are among the most widely used inorganic carriers, typically employed as beads, gels, or nanoparticles.
- Silica/Porous Glass: These kinds provide a large surface area for the enzyme to attach to the carrier. Mesoporous silica (with pore sizes of 2-50 nm allows for high loading capacity.
Their rigidity and resistance to crushing or deformation are crucial for maintaining stable pore diameter and volume in the challenging environment of industrial packed-bed and stirred-tank reactors.
Silica has high operational stability since it is chemically stable; the surface’s natural silanol (Si-OH) groups allow for easy modification through silanization chemistry.
Ceramics and Metal Oxides: This category encompasses carriers like alumina (Al2O3), iron oxides (Fe3O4), titania (TiO2), and zirconia (ZrO2). These materials are highly valued for applications demanding extreme temperature or pressure tolerance.
Ceramics offer outstanding mechanical strength and resistance to attrition, allowing for long operational life in industrial reactors.
Similar to silica, Al2O3 can be synthesized with high porosity and controllable pore sizes to optimize loading and mass transfer.
Al2O3 maintains its structure and integrity at very high temperatures and under harsh chemical conditions (strong acids/bases), making it ideal for high-temperature catalytic reactions.
- Organic/Polymeric Carriers (Flexible and Functional):
- Natural Polymers: Being derived from biological sources such as Alginate, Agar, and Cellulose, are highly favoured for applications, particularly in biomedicine and food technology
Typically, non-toxic and well-tolerated by living systems (cells, organisms), the biocompatibility of natural polymers is essential for pharmaceutical, food, and environmental applications. They can be broken down easily, reducing environmental impact and aiding in controlled release systems.
These are easily modifiable since they contain numerous natural functional groups, primarily hydroxyl (OH) and amino (NH2) groups. Many form hydrogels under mild conditions (e.g., room temperature, neutral pH). Preserves the native structure and activity of sensitive biomolecules like enzymes.
- Synthetic Polymers:
Synthetic polymers are widely used for enzyme immobilization due to their superior chemical tunability, structural adaptability, and mechanical durability.
These polymers are reusable and can be tailored according to the requirement; this functionality gives them an edge. They offer strong, durable support, allowing them to withstand continuous operation despite chemical and physical stresses like high flow rates, extreme pH, or organic solvents.
This resilience is key to preventing the enzyme from degrading or escaping (leaching) from the support. Poly (methyl methacrylate) (PMMA), Polyacrylamide (PAM), Vinyl and Styrene-Based Polymers are some of the highly used options for synthetic polymers used in immobilization.
Ideal Carrier Properties:
- High Mechanical Strength/Durability
- Optimal Particle Size and Shape
- Chemical and Thermal Stability
- Regenerability (removal of the deactivated enzyme and the subsequent re-immobilization of fresh enzyme)
- Low Swelling (material should not swell or shrink significantly in different buffers or organic solvents, as this can destroy the matrix structure and impede flow)
- Appropriate Pore Size (for efficient diffusion)
- Easily Modifiable/Reactive Groups
- Biocompatibility/non-toxicity
- Low Cost and Wide Availability
- Ease of Separation
- Non-Compressibility
Enzyme immobilization is a transformative technology that directly addresses the inherent limitations of using fragile, high-cost, soluble enzymes in industrial settings. By anchoring these powerful protein biocatalysts, whose efficiency is dictated by their complex structure and turnover rate onto diverse and strategically chosen inorganic or organic carrier materials, the process achieves its primary goal: transitioning enzymes from a delicate reagent to a robust, reusable, and cost-effective catalyst.
Historically validated by commercial successes like the industrial production of L-amino acids, the field continues to advance through the engineering of carriers with tailored properties (high stability, optimal pore size, and mechanical strength). Ultimately, enzyme immobilization serves as a vital bridge, enabling the full-scale application of biology’s most efficient catalysts in sectors ranging from pharmaceuticals and food to bioremediation, thereby driving sustainable and highly efficient manufacturing processes. The strategic choice of carrier remains the critical factor in unlocking the enzyme’s optimal, long-term industrial performance.
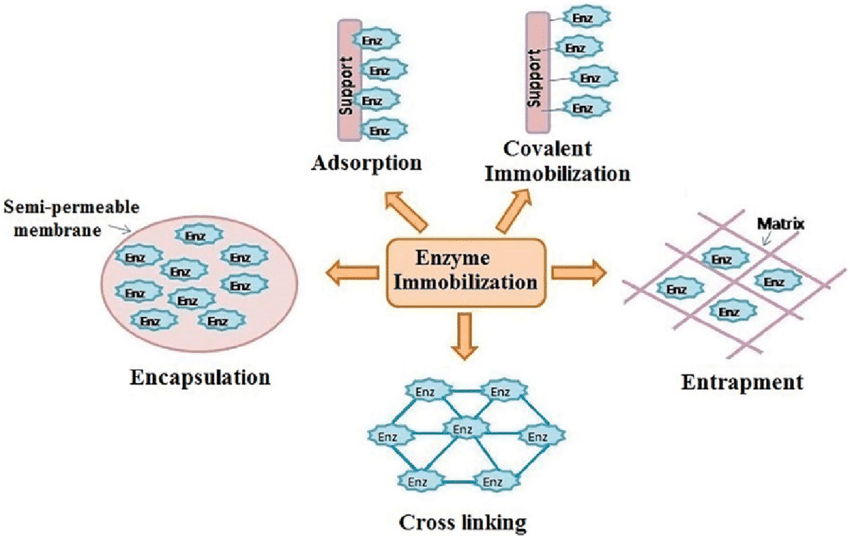
Immobilization methods are broadly classified based on the nature of the binding (physical or chemical) between the enzyme and the support material. The support material, or carrier, is the inert phase used to anchor the enzyme. A wide variety of materials are used, like organic, inorganic, organic synthetic, etc. The choice of technique and carrier is specific to the enzyme, the substrate, and the desired industrial application.
Physical Methods (Weaker, Reversible Interactions)
- Adsorption:
Adsorption is often used as a preliminary method when it comes to enzyme immobilization. It relies on non-covalent, reversible interactions between the enzyme molecule and the surface of a support material. The enzyme binds to the outer surface of a carrier (the support material) through weak, non-covalent forces such as Van der Waals Forces, Hydrogen Bonds, and Ionic Interactions. The procedure is very straightforward, typically involving only mixing the enzyme solution with the carrier, often requiring just stirring or incubation. Because the weak binding doesn’t significantly alter the enzyme’s three-dimensional structure, the initial catalytic activity is often preserved at a high level. In some cases, the enzyme can be easily desorbed, and the carrier can be regenerated or reused.
The carrier is washed to make the surface clean and ready for binding
Enzyme is dissolved into a buffer solution, and pH is often chosen to be close to the isoelectric point (pI) of the enzyme
The carrier is mixed with the enzyme solution. The mixture is gently stirred for a specific period under optimum conditions.
During this time, the enzyme molecules diffuse from the solution and physically adhere to the available surface area of the carrier
Examples: Lipase Immobilization on Hydrophobic Supports, Glucose Isomerase (GI) on Ion-Exchange Resins, Urease on Carbon Materials
- Entrapment/Inclusion:
Entrapment is a method where the enzyme molecules are physically locked within the structure of a matrix, such as a porous polymer lattice or capsule, without being chemically bonded to the material. The core principle is the formation of a stable, porous network around the enzyme. The enzyme solution is mixed with a monomer or polymer solution, and then a quick polymerization or cross-linking reaction is initiated. As the polymer solidifies (forming a gel or fibre), the large enzyme molecules are trapped within the microscopic lattice structure (like being caught in a net or cage).
The pore size of the resulting matrix is carefully controlled. It must be small enough to prevent the larger enzyme molecules from diffusing out (preventing leakage) but large enough to allow the smaller substrate molecules to diffuse in and the product molecules to diffuse out.
Unlike chemical binding, entrapment is mild and doesn’t require direct reaction with the enzyme’s active site, making it ideal for immobilizing entire microbial cells or complex multi-enzyme pathways.
Examples: Alginate, Carrageenan, Polyacrylamide Gel (PAM)
https://www.researchgate.net/figure/Different-methods-of-enzyme-immobilization_fig4_336759040
- Encapsulation/Containment:
Encapsulation, or containment, is a method in which enzymes are enclosed within a distinct compartment, typically defined by a semipermeable membrane or thin polymeric shell.
The fundamental principle is to create a boundary or barrier around the enzyme, trapping it in a concentrated, internal volume. It is typically done by forming a thin, semipermeable polymer shell around a concentrated enzyme solution. This creates microcapsules or vesicles that trap the enzyme inside while allowing small substrates and products to pass through the membrane.
Enzymes are in the aqueous core; two reactive monomers, starting in separate immiscible liquids, rapidly react at the droplet interface to form a polymer shell (e.g., Nylon microcapsules).
Chemical Methods (Stronger, Permanent Interactions)
- Covalent Bonding (The Gold Standard):
A covalent bond is the strongest bond due to the sharing of electrons to attain a stable electronic configuration. A covalent bond creates a strong link between the carrier and the enzyme.
The enzyme provides nucleophilic functional groups, which are typically the side chains of amino acid residues that are not critical for the active site’s function. The inert carrier (e.g., agarose, silica, polymer beads) is typically chemically activated to introduce highly electrophilic groups that can readily react with the enzyme’s groups.
The reaction occurs in two sequential steps. The solid support material is chemically treated (for example, using CNBr or glutaraldehyde) to introduce highly reactive groups (anchors) onto its surface. This makes the typically inert carrier ready to react with the enzyme. The enzyme is then introduced, and the reactive groups on the activated carrier form a strong, permanent covalent bond (such as an amide, ether, or imine bond) with available nucleophilic groups on the enzyme’s structure.
The main goal is to create an immobilization matrix that exhibits zero leaching (no enzyme detachment), providing superior operational stability and the potential for repeated use in industrial biocatalysis
- Cross-Linking (Carrier-Free Immobilization):
Cross-linking involves using a small, multi-functional molecule to chemically stitch individual enzyme molecules together, forming a solid, insoluble particle.
Bifunctional reagents such as glutaraldehyde or diazonium salts have two reactive groups at opposing ends. They act as molecular glue. The enzyme is typically first precipitated (CLEAs, Cross-Linked Enzyme Aggregates) or crystallized (CLECs, Cross-Linked Enzyme Crystals) to bring the molecules into proximity. The bifunctional reagent is added. One reactive end links to a functional group (like a lysine -NH2 group) on one enzyme molecule, and the other reactive end links to a functional group on an adjacent enzyme molecule. This process is repeated across thousands of molecules, forming a stable, macroscopic structure.
Since there is no inert carrier material (the structure is 100% enzyme), the concentration of active enzyme per unit mass is maximized.
CLEA/CLEC formation works best with highly purified enzymes. The presence of contaminating proteins can lead to their inclusion in the cross-linked structure, potentially wasting reagents and altering the particle properties
Industrial and Emerging Applications
- Bioremediation and Environmental Uses:
- Immobilized enzymes are crucial for bioremediation and environmental detoxification. By fixing enzymes like peroxidases and laccases onto a stable support, they can be efficiently used to degrade persistent organic pollutants such as pesticides and phenols found in wastewater.
- Food and Beverage Industry:
- The production of High-Fructose Corn Syrup (HFCS) relies entirely on immobilized glucose isomerase to convert glucose into sweeter fructose.
- Enzyme Lactase (beta-galactosidase) is responsible for the hydrolysis of lactose into more digestible monosaccharides, glucose, and galactose. A breakthrough for individuals suffering from lactose intolerance.
- Biosensors and Diagnostics:
- Immobilized enzymes serve as the core bioreceptors in biosensors, achieving high selectivity by catalyzing a reaction on a target analyte (e.g., glucose). The immobilization fixes the enzyme to the transducer, where the resulting chemical change (e.g., H2O2 production) is immediately converted into a measurable electrical signal. This process ensures the sensor is stable, reusable, and highly accurate for diagnostics.
- Pharmaceutical and Fine Chemical Synthesis:
- The primary use of immobilized enzymes in this sector is for stereospecific synthesis (or chiral synthesis), which is essential for producing pure drug molecules.
Current Challenges and Future Outlook
Operational Hurdles
- Immobilization can cause steric hindrance, leading to a significant loss of enzyme activity.
- Resistance to the movement of substrates and products limits reaction kinetics, reducing overall efficiency
- Maintaining long-term mechanical stability in continuous industrial reactors is a persistent challenge.
Future Directions
- Site-directed immobilization technique can be used to bind the targeted residue away from the enzyme’s active site, which would be a predetermined location
- Utilizing artificial intelligence (AI) for the rational design of advanced carriers and methods.
- Developing stimuli-responsive immobilized enzyme systems that can be controlled by external triggers (e.g., pH, temperature).
References
- Homaei, A. A., Sariri, R., Vianello, F., & Stevanato, R. (2013). Enzyme immobilization: an update. Journal of Chemical Biology, 6(4), 185–205. https://doi.org/10.1007/s12154-013-0102-9
- Bata, G. H., El-Khatib, E. M., Aboelwafa, A. H., & Abo-Elella, S. M. (2023). Enzyme Immobilization Technologies and Industrial Applications. ACS Omega, 8(10), 9197–9212. https://doi.org/10.1021/acsomega.2c0756 Enzyme immobilization.pdf
- Ahmad, R., & Sardar, M. (2015). Enzyme Immobilization: An Overview on Methods, Support Material, and Applications of Immobilized Enzymes. In M. R. K. Z. Idrus, M. M. I. Idrus, & I. K. E. A. Z. W. A. M. Abdullah (Eds.), New Perspectives in Biosensors and Biodetection
- Nguyen, H. H., & Kim, M. (2017). An Overview of Techniques in Enzyme Immobilization. Applied Science and Convergence Technology, 26(6), 157–169. https://doi.org/10.5757/ASCT.2017.26.6.157
- Sheldon, R. A. (2007). Enzyme immobilization: The quest for optimum performance. Advanced Synthesis & Catalysis, 349(8–9), 1289–1307.
- Abdelmajeed, N. A., Khelil, O. A., & Danial, E. N. (2012). Immobilization technology for enhancing bio-products industry. African Journal of Biotechnology, 11(73), 13528–13539. https://doi.org/10.5897/AJB12.547
- National Human Genome Research Institute. (n.d.). Enzyme. Talking Glossary of Genomic and Genetic Terms. Retrieved November 10, 2025, from https://www.genome.gov/genetics-glossary/Enzyme
- Robinson, P. K. (2015). Enzymes: Principles and biotechnological applications. Essays in Biochemistry, 59, 1–41. https://doi.org/10.1042/bse0590001
- “Biochemistry, Proteins Enzymes.” StatPearls, StatPearls Publishing, 2025, www.ncbi.nlm.nih.gov/books/NBK554481/.
- Khan, M. R. (2021). Immobilized enzymes: a comprehensive review. Bulletin of the National Research Centre, 45(1), 207. https://doi.org/10.1186/s42269-021-00649-0
- Robescu, M. S., & Bavaro, T. (2025). A comprehensive guide to enzyme immobilization: All you need to know. Molecules, 30(4), 939. https://doi.org/10.3390/molecules30040939
- Boudrant J., Woodley J.M., Fernandez-Lafuente R. Parameters Necessary to Define an Immobilized Enzyme Preparation. Process Biochem. 2020;90:66–80. doi: 10.1016/j.procbio.2019.11.026.
- Wu S., Snajdrova R., Moore J.C., Baldenius K., Bornscheuer U.T. Biocatalysis: Enzymatic Synthesis for Industrial Applications. Angew. Chem. Int. Ed. 2021;60:88–119. doi: 10.1002/anie.202006648.
- https://doi.org/10.1002/adsc.200700082
- https://www.youtube.com/watch?v=sL_iEOuvK80
- https://www.youtube.com/watch?v=oVJ2LJxO6tU
- https://www.youtube.com/watch?v=h1Q-_sL_2kA
- https://www.youtube.com/watch?v=epIf2t2ntPA
- https://www.youtube.com/watch?v=sL_iEOuvK80
